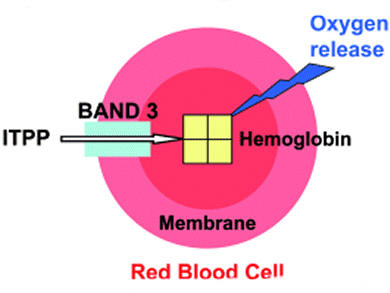
Numerous diseases, such as cardiovascular ailments and cancer, are characterized by a lack of oxygen in specific tissues. Therefore, increasing the supply of oxygen delivered by red blood cells (RBCs) to counteract the effects of hypoxia has much potential therapeutic interest. Nobel laureate Jean-Marie Lehn and colleagues at the Université de Strasbourg, France, have undertaken a series of collaborative research efforts to address this.
Oxygen release from hemoglobin (Hb) is regulated in humans by the allosteric effector 2,3-bisphosphoglycerate (2,3-BPG). Increased oxygen release may in principle be achieved by a more powerful effector molecule that would bind Hb, shift its oxygenation curve, and be able to cross the RBC plasma membrane. It was discovered earlier that myo-inositol trispyrophosphate (ITPP) is such a molecule, and is able to markedly increase oxygen delivery by RBCs both ex vivo and in vivo. It is now found (ChemBioChem, referenced below) that ITPP entry into RBCs is mediated by the anion transporter Band 3 protein and may be suppressed by specific inhibition of this carrier. As Band 3 is mainly localized in the RBC membrane, the uptake of ITPP is highly tissue specific towards RBCs, a feature of major importance for its potential medical use. The therapeutic value of such effector molecules warrants a broad exploration of the chemical space, involving both molecular recognition of the allosteric pocket of Hb and oxygen release from Hb.
The synthesis of a number of phosphorylated derivatives of hexapyranoses [1] and of selectively substituted myo-inositol derivatives [2], together with the investigation of their effect on oxygen release from pure Hb allow the exploration of structure-activity relationships for various molecular features (such as charge and shape) and provides information about the design of allosteric effectors of Hb as potential drugs that present a wide range of activities.
- myo-Inositol Trispyrophosphate: A Novel Allosteric Effector of Hemoglobin with High Permeation Selectivity across the Red Blood Cell Plasma Membrane,
Jean-Marie Lehn
ChemBioChem 2010, 11 (18), 2543–2548
DOI: 10.1002/cbic.201000499 - [1] http://doi.org/10.1002/cmdc.201000366
- [2] http://doi.org/10.1002/cmdc.201000421