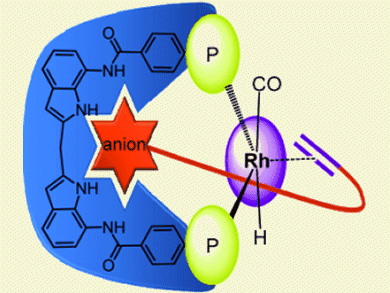
The application of a supramolecular strategy for the preorganization of alkene substrates for selective rhodium catalyzed hydroformylation is described by Joost N. H. Reck and colleagues, Van’t Hoff Institute for Molecular Sciences, University of Amsterdam, The Netherlands. They introduced a new bidentate phosphorus ligand with an integral anion-recognition site. The ligand helps preorganize alkenes that contain anionic functionalities prior to hydroformylation.
Calculations showed that small substrates, such as the 3-butenoate anion were too small to bind in the pocket, while its double bond was coordinated to the metal center. Larger substrates such as the 4-pentenoate anion could be bound as they easily spanned the distance between metal and binding site.
For the substrates which were too small to bind, the linear/branched ratio of hydroformylation products ranged between 1.6 and 2.6. In contrast, the 4-pentenoate ion was hydroformylated with unprecedented selectivity for the linear aldehyde (l/b ratio of 40).
- Remote Supramolecular Control of Catalyst Selectivity in the Hydroformylation of Alkenes,
Paweł Dydio, Wojciech I. Dzik, Dr. Martin Lutz, Bas de Bruin, Joost N. H. Reek,
Angew. Chem. Int. Ed. 2011, 50, 396.
DOI: 10.1002/anie.201005173