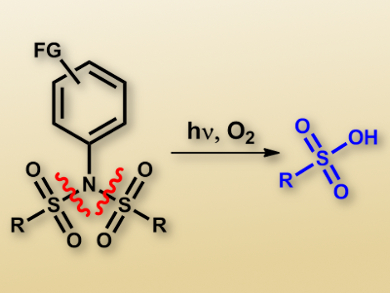
Photoacid generators (PAGs) are important tools in materials science. With application possibilities ranging from nanoelectronics, such as in photoresists and semiconductor materials, to materials used for coating or curing, there is a demand for a broad range of photoacids and photoacid generators. It is known that ionic PAGs, such as those based on sulfonium or iodonium salts, can be excellent in their PAG capabilities. However, it is also useful to create nonionic PAGs, as ionic compounds can create problems for their implementation in applications due to, for example, limited solubility.
Maurizio Fagoni and colleagues, University of Pavia, Italy, have investigated a new class of nonionic PAGs which is able to release up to two equivalents of strong sulfonic acids (methanesulfonic and p-toluenesulfonic acids) for each mole of photoactive substrate upon irradiation. These compounds, differently substituted N-arylsulfonimides, were efficiently obtained in a straightforward way from the corresponding commercially available anilines, and their absorption properties and photochemical behavior could be simply tuned by choosing suitable substituents.
In addition, the researchers investigated the mechanism of the obtained photochemical reactivity, which is a crucial factor for the further development of better PAG systems. According to the proposed mechanism, irradiation of the compounds causes excitation to a singlet state, which is followed by homolytic fragmentation of one of the N−S bonds. The formed radicals can follow two competing pathways, namely, recombination to yield Fries rearrangement products or hydrogen abstraction from the reaction medium.
To test the potential of the investigated compounds as PAGs, one of the sulfonimides was used to promote the acid-induced gelation of aqueous (2 wt %) alginate polymer. The light-induced acid release promoted gelation under irradiation at 310 nm.
- Photochemistry of N-Arylsulfonimides: An Easily Available Class of Nonionic Photoacid Generators (PAGs),
Edoardo Torti, Stefano Protti, Daniele Merli, Daniele Dondi, Maurizio Fagnoni,
Chem. Eur. J. 2016.
DOI: 10.1002/chem.201603522