Lithium-ion batteries with a Li metal anode are considered a strong candidate for the next generation of rechargeable batteries because of their high theoretical capacity (3860 mAh g–1). Unfortunately, the high volume change of Li and the formation of dendrites upon cycling pose a safety risk and make this type of battery rather short-lived in operation.
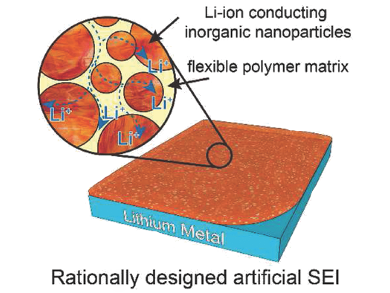
Yi Cui, Stanford University, USA and colleagues have developed an artificial solid electrolyte interphase (SEI) that forms a protective layer on Li metal anodes to meet these challenges. First, CuN3 nanoparticles (NPs) were prepared through the reaction of Cu(OMe)2 with benzylamine and subsequently dispersed in THF together with the polymer styrene butadiene rubber (SBR). The dispersion was then either drop-cast or blade-coated onto the metal anode where the NPs were immediately passivated by the formation of LiN3 – one of the fastest Li-ion conductors.
The resulting coating efficiently supressed the formation of dendrites and maintained integrity during cycling, which was demonstrated by a high coulombic efficiency of more than 97.4 %. Furthermore, the researchers successfully demonstrated that their approach can be used to homogenously coat novel porous Li metal anodes to further reduce the detrimental effects during cycling.
- An Artificial Solid Electrolyte Interphase with High Li-Ion Conductivity, Mechanical Strength, and Flexibility for Stable Lithium Metal Anodes,
Yayuan Liu, Dingchang Lin, Pak Yan Yuen, Kai Liu, Jin Xie, Reinhold H. Dauskardt, Yi Cui,
Adv. Mater. 2016.
DOI: 10.1002/adma.201605531