Sulfur is a cheap and abundant element. Lithium-sulfur batteries could serve as a high-capacity alternative to the commonly used lithium-ion batteries. However, until now such batteries have problems with cycling stability and with the poor electric conductivity of sulfur. Incorporating sulfur into carbon nanomaterials could lead to improved cathode materials for lithium-sulfur batteries.
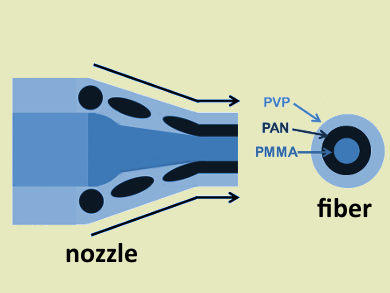
Arumugam Manthiram, University of Texas at Austin, USA, and colleagues have developed a synthesis of porous carbon nanotubes containing immobilized sulfur. The team used a dual-nozzle electrospinning process to create three-layered polymer nanofibres. They used an inner nozzle to generate a poly(methylmethacrylate) (PMMA) core and an outer nozzle to prepare poly(acrylonitrile) (PAN) and poly(vinylpyrrolidone) (PVP) coatings (pictured).
The polymer nanofibres were then carbonized and activated with KOH. Only the PAN tube (pictured in black) is transformed into graphitic carbon during this process, thus creating porous carbon nanotubes (CNTs). The researchers immobilized sulfur on the CNTs using a sodium thiosulfate solution and hydrochloric acid.
The resulting composite material was used as a cathode for Li–S batteries and has an excellent cycle stability (917 mAh/g after 300 cycles) and rate capability (859 mAh/g at 5 C). The porous structure allows for a high sulfur loading of 4.6 mg cm−2. According to the team, this strategy could solve the problems in practical Li–S battery applications.
- Sulfur-Immobilized, Activated Porous Carbon Nanotube Composite Based Cathodes for Lithium-Sulfur Batteries,
Jun Seop Lee, Jaemoon Jun, Jyongsik Jang, Arumugam Manthiram,
Small 2017.
DOI: 10.1002/smll.201602984