Replacing expensive noble metal catalysts with earth-abundant 3d metals has advantages such as lower cost and improved sustainability and could lead to the discovery of new reactivity. While catalysts based on metals such as iron, cobalt, and copper have been extensively investigated, less is known about manganese-mediated reactions.
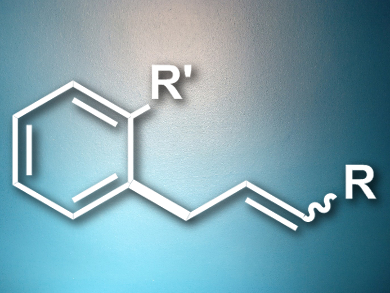
Frank Glorius, University of Münster, Germany, and colleagues have developed a manganese-catalyzed allylation protocol which combines C–H and C–C/C–X bond activation. The team reacted a range of aromatic substrates with different olefins in the presence of [MnBr(CO)5] to give allylic alcohols, allylated arenes, cyclopentenes, and 1,4-dienes in good to excellent yields (example pictured). Based on evidence from radical trapping experiments and H/D scrambling experiments, the team proposes a mechanism involving a base-assisted cyclomanganation of the substituted arene, followed by coordination and insertion of the alkene and protonolysis to give the final product.
The reactions could be performed under solvent-free conditions, which represents the first example of 3d metal-catalyzed C–H activation under neat conditions without a large excess of one coupling partner acting as the solvent. According to the researchers, the developed reaction protocol can be readily scaled up and expands the scope of manganese catalysis.
- Manganese-catalyzed allylation via sequential C–H and C–C/C–Het bond activation,
Qingquan Lu, Felix J. R. Klauck, Frank Glorius,
Chem. Sci. 2017.
DOI: 10.1039/c7sc00230k