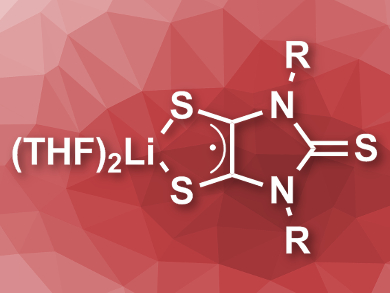
Metal–dithiolene complexes are found in enzymes, and synthetic variants can be used, e.g., in optoelectronics. Dithiolenes are so-called “non-innocent” ligands, which means they are redox-active and can change the oxidation state of the metal center. The ligand has radical character in this case. The corresponding anionic free radicals are highly reactive and had so far only been studied computationally, spectroscopically, or in low-temperature matrices.
Gregory H. Robinson, University of Georgia, Athens, USA, and colleagues have synthesized and characterized the lithium salt of a stable anionic dithiolene radical. The team activated an N-heterocyclic carbene at the C4 position using lithiation and reacted the activated carbene with elemental sulfur to create a disulfurized derivative. A further reaction with elemental sulfur gives the desired lithium salt of the dithiolene radical (pictured). In the solid state, the product is stable indefinitely under an argon atmosphere.
The team characterized the dithiolene radical using 1H NMR, UV-Vis, and electron paramagnetic resonance (EPR) spectroscopy, as well as X-ray crystallography. The compound’s use as a ligand was also studied, and it was found to form an anionic chlorogermanium-bis(dithiolene) complex when combined with GeCl2·dioxane in tetrahydrofuran (THF). According to the researchers, the synthesized radical could allow the synthesis of other so far unexplored dithiolene compounds.
- A Stable Anionic Dithiolene Radical,
Yuzhong Wang, Hunter P. Hickox, Yaoming Xie, Pingrong Wei, Soshawn A. Blair, Michael K. Johnson, Henry F. Schaefer, Gregory H. Robinson,
J. Am. Chem. Soc. 2017.
DOI: 10.1021/jacs.7b03794