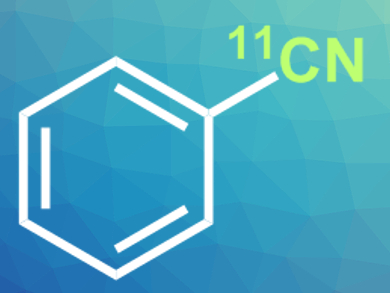
Positron emission tomography (PET) scans are used in medicine to observe biochemical processes in the body. They are used, for example, to detect cancer metastasis, but also to study brain activity or inflammations. To make these processes visible, radiotracers are used.
Radiotracers are biologically active molecules, e.g., glucose or amino acid derivatives, labeled with radionuclides such as 11C or 18F. To create 11C-labeled compounds, methylation reactions are used most often. The development of other labeling approaches could provide new and useful tracers.
Neil Vasdev, Steven H. Liang, Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA, and colleagues have developed a synthesis approach for aromatic 11C-nitriles (example pictured). The team performed the cyanation of arylboronic acids with carbon-11-labelled CsCN, using a CuI as a copper(I) catalyst, cesium carbonate as a base, and N,N‘-dimethylethylenediamine (DMEDA) as an additive.
The reaction provides good yields, has a broad substrate scope, and the resulting 11C-nitriles can be further converted into amides, amines, or carboxylic acids.The researchers hope to use this approach for the synthesis of a range of 11C-labeled radiotracers.
- [11C]Cyanation of arylboronic acids in aqueous solutions,
Longle Ma, Michael S. Placzek, Jacob M. Hooker, Neil Vasdev, Steven H. Liang,
Chem. Commun. 2017.
DOI: 10.1039/c7cc02886e