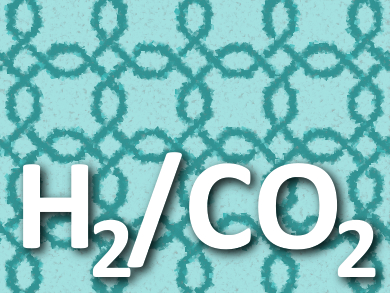One approach to reduce CO2 emissions is the decarbonization of fossil fuels. Here, the fossil fuels are converted to H2 and CO2 at high temperatures and under high pressures. CO2 is then separated and sequestered and only H2 is used further. For this method, materials that can selectively and efficiently separate the two gases are needed.
Haiqing Lin and colleagues, University at Buffalo, The State University of New York, USA, have developed an acid-doped polymer membrane with an unprecedented H2/CO2 selectivity. The team immersed films of poly[2,20-(m-phenylene)-5,5-bibenzimidazole] (PBI) in solutions of H3PO4 or H2SO4. The acid molecules act as hydrogen-bonded bridges between imidazole rings in the polymer. This causes crosslinking between the polymer chains and improves the chain-packing efficiency.
The researchers used the synthesized crosslinked polymer membranes for H2/CO2 separation. They found that the H3PO4-doped membrane has an H2/CO2 selectivity of 140 at 150 °C, an unprecedented value for polymeric materials and one that is comparable to ordered 2D materials such as graphene oxide or metal-organic frameworks.
- Unprecedented size-sieving ability in polybenzimidazole doped with polyprotic acids for membrane H2/CO2 separation,
Lingxiang Zhu, Mark T. Swihart, Haiqing Lin,
Energy Environ. Sci. 2018.
https://doi.org/10.1039/c7ee02865b