Anna Walczak Adam Mickiewicz University in Poznań, and Artur R. Stefankiewicz, Centre for Advanced Technologies, Poznań, Poland, have synthesized pH-responsive metallosupramolecular Pd(II) complexes. A pH change reversibly changes the morphology, composition, charge, and optical properties of the complexes.
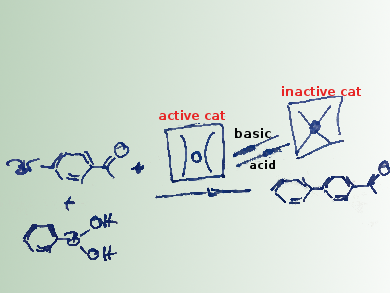
The team has used this mechanism to create a catalytic system in which each of the two isomers presents distinct catalytic activities in the Suzuki–Miyaura reaction. Under the normal basic conditions for a Suzuki–Miyaura coupling, only the diketonate forms are present. They act as very efficient catalysts for this reaction. The complexes are air-stable crystalline solids, so a simple reaction setup, without excluding air or water, and efficient catalysis under mild conditions are possible. In addition, the catalytic process can be quenched (by the addition of an a cid) and reactivated (by the addition of a base) at any stage without the risk of losing catalytic activity due to the dynamic change of the complexes.
According to the researchers, this type of externally addressable dynamic supramolecular system could play a major role in the development of “smart” functional nanostructures.
- pH-Induced Linkage Isomerism of Pd(II) Complexes: A Pathway to Air- and Water-Stable Suzuki–Miyaura-Reaction Catalysts,
Anna Walczak, Artur R. Stefankiewicz
Inorg. Chem. 2017.
https://doi.org/10.1021/acs.inorgchem.7b02711