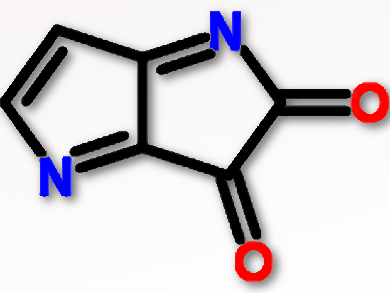
Conjugated donor-acceptor (D-A) polymers are of interest for solution-processable semiconductors with high charge mobility for applications such as E-papers and wearable electronics. Diketopyrrolopyrrole (DPP; pictured), which has two flanking thiophenes, is often used as an electrode acceptor in combination with various electron donors to form D-A polymers. To increase solubility in organic solvents, two bulky branching alkyl groups can be incorporated. However, these groups affect the planar geometry of the conjugated polymer backbone and prevent dense packing of polymer chains which restricts charge mobility.
Deqing Zhang, University of Chinese Academy of Sciences, Beijing, China, and colleagues have developed two new DPP-selenophene based conjugated polymers (PDPPSe), PDPPSe-10 and PDPPSe-12, with improved semiconducting performances in which one of the two bulky branching alkyl chains is replaced by the linear groups n-decyl and n-dodecyl, respectively. Due to a reduction in steric hindrance, the conjugated backbone of both PDPPSe-10 and PDPPSe-12 became more planar which improved packing and π-electron delocalization resulting in a lower band gap energy.
The researchers fabricated polymer films of both PDPPSe-10 and PDPPSe-12 by spin-coating. When compared to bulky PDPPSe, the PDPPSe-10 and PDPPSe-12 showed more ordered interchain packing and thin film crystallinity. These overall improvements lead to a remarkable enhancement of the charge mobility. When integrated into field-effect transistors (FETs), PDPPSe-10 and PDPPSe-12 showed improved hole mobilities of 8.1 and 9.4 cm2V–1S–1, respectively, compared to PDPPSe (1.35 cm2V–1S–1). Under nitrogen atmosphere, both PDPPSe-10 and PDPPSe-12 behave as ambipolar semiconductors with increased hole/electron mobilities of 6.5 and 7.9 cm2V–1S–1, respectively.
- Charge Mobility Enhancement for Conjugated DPP-Selenophene Polymer by Simply Replacing One Bulky Branching Alkyl Chain with Linear One at Each DPP Unit,
Zhijie Wang, Zitong Liu, Lu Ning, Mingfei Xiao, Yuanping Yi, Zhengxu Cai, Aditya Sadhanala, Guanxin Zhang, Wei Chen, Henning Sirringhaus, Deqing Zhang,
Chem. Mater.2018.
https://doi.org/10.1021/acs.chemmater.8b01007