Nitric oxide (NO) is a toxic gas, but also acts as an important signaling molecule in biology. It causes, e.g., the dilation of blood vessels, which is used to treat cardiac disease. NO also has antimicrobial properties and can prevent blood clots. However, the compound is very reactive and, thus, usually needs to be generated in situ from more stable precursors. NO-releasing polymers, for example, could be used in medical devices to prevent clots and infections.
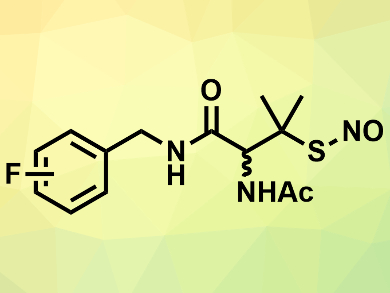
Mark E. Meyerhoff, University of Michigan, Ann Arbor, USA, and colleagues have synthesized a range of fluoro S-nitrosothiols, (example pictured), which could act as NO donors. The compounds were synthesized from the corresponding fluorinated benzylamines and 3-acetamido-4,4-dimethylthietan-2-one via a nucleophilic ring opening reaction. The phenyl ring acts as a spacer to reduce the electron-withdrawing effects of the fluoro substituents on the relatively weak S–N bond.
The prepared fluoro S-nitrosothiols release NO in stoichiometric quantities under irradiation with light and when heated. They are converted to their corresponding disulfides in the process. According to the researchers, the compounds could be incorporated into fluorinated polymers for use in medical devices.
- Synthesis and nitric oxide releasing properties of novel fluoro S-nitrosothiols,
Yang Zhou, Jinyi Tan, Yuping Dai, Yanmin Yu, Qi Zhang, Mark E. Meyerhoff,
Chem. Commun. 2019.
https://doi.org/10.1039/c8cc08868c