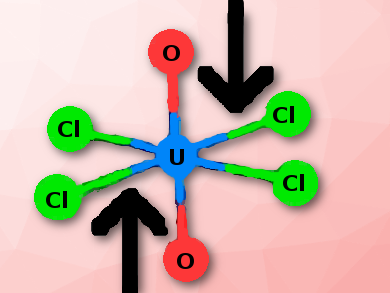
Most proposed methods of storing radioactive waste involve burying it deep underground, often in a salt formation. Eventually, the salt will collapse around the waste stored there and will place it under pressures. To understand the effects of pressure on the chemistry of the stored material, Thomas E. Albrecht-Schmitt and colleagues, Florida State University, Tallahassee, USA, have studied the pressure behavior of crystalline Cs2UO2Cl4 using a diamond anvil cell. To magnify the effects pressure has, they used higher pressure over a much shorter period of time than in a collapsed salt formation.
U(VI) in the form of the uranyl ion (UO22+) is the most environmentally stable form of uranium under oxidizing conditions. Its ambient pressure chemistry has been well studied spectroscopically and computationally, however, no experimental studies on the pressure behavior of its molecular complexes have been reported.
The team observed with increasing pressure a dramatic decrease the uranyl fluorescence intensity and an apparent linear decrease in bond length using the O−U−O symmetric stretching frequency. Evidence for the stabilization of the uranyl bond was found and it is likely that the uranyl ion is less reactive under pressure.
As both the radiative rate and energy of fluorescence remain unchanged, the decrease in fluorescence intensity with increasing pressure was attributed entirely to increased relaxation through phonon modes. Contrary to earlier DFT calculations, quantum theory of atoms in molecules (QTAIM) calculations and ab initio wave function methods (CASSCF) support the suggestion that the U−O bond is strengthened by pressure in a way that suggests a linear pressure−bond distance relationship. DFT overestimates the covalency of the U−Cl bond and its effect on the U−O bond.
- High-Pressure Studies of Cesium Uranyl Chloride,
Evan Warzecha, Cristian Celis-Barros, Tristan Dilbeck, Kenneth Hanson, Thomas E. Albrecht-Schmitt,
Inorg. Chem. 2018.
https://doi.org/10.1021/acs.inorgchem.8b02300