Tetsuya K. Sato, Japan Atomic Energy Agency (JAEA), Tokai, Japan, and international colleagues have determined the first ionization potentials of the artificially created elements fermium (Fm, atomic number Z = 100), mendelevium (Md, Z = 101), nobelium (No, Z = 102), and lawrencium (Lr, Z = 103). The data show that the actinide series ends with lawrencium. The data agree well with those predicted by state-of-the-art relativistic calculations.
The elements 100 to 103 do not occur naturally on earth. They are artificially created in nuclear fusion reactions at particle accelerators. The researchers used the Tandem accelerator and the attached isotope separator at JAEA. The first ionization potentials were determined using a surface ionization process. A gas stream in a Teflon tube carries the elements to a tantalum chamber with a surface heated to up to 3,000 °C, where they can be ionized. Comparing the number of atoms fed in with that of ionized atoms provides a value for the efficiency of the ionization. From this, the first ionization potential of the elements can be determined.
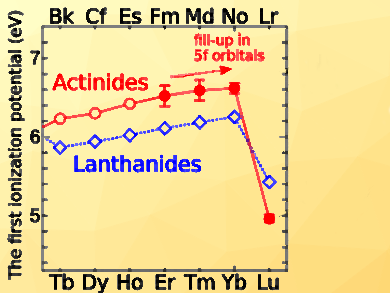
The first ionization potentials of the elements measure the energy required to remove the least tightly bound electron from the outer shell of a neutral atom. The ionization potentials increase up to No. Lr, the following element, showed a decrease in the ionization potential. These results demonstrate that the 5f orbital is fully filled at No with the [Rn]5f147s2 configuration and that Lr has a weakly bound electron outside the No core ([Rn]5f146d7s2). These results verify that the actinide series ends with Lr.
- First Ionization Potentials of Fm, Md, No, and Lr: Verification of Filling-Up of 5f Electrons and Confirmation of the Actinide Series,
Tetsuya K. Sato, Masato Asai, Anastasia Borschevsky, Randolf Beerwerth, Yusuke Kaneya, Hiroyuki Makii†, Akina Mitsukai, Yuichiro Nagame, Akihiko Osa, Atsushi Toyoshima, Kazuaki Tsukada, Minoru Sakama, Shinsaku Takeda, Kazuhiro Ooe, Daisuke Sato, Yudai Shigekawa, Shin-ichi Ichikawa, Christoph E. Düllmann, Jessica Grund, Dennis Renisch, Jens V. Kratz, Matthias Schädel×, Ephraim Eliav, Uzi Kaldor, Stephan Fritzsche, Thierry Stora,
J. Am. Chem. Soc. 2019.
https://doi.org/10.1021/jacs.8b09068