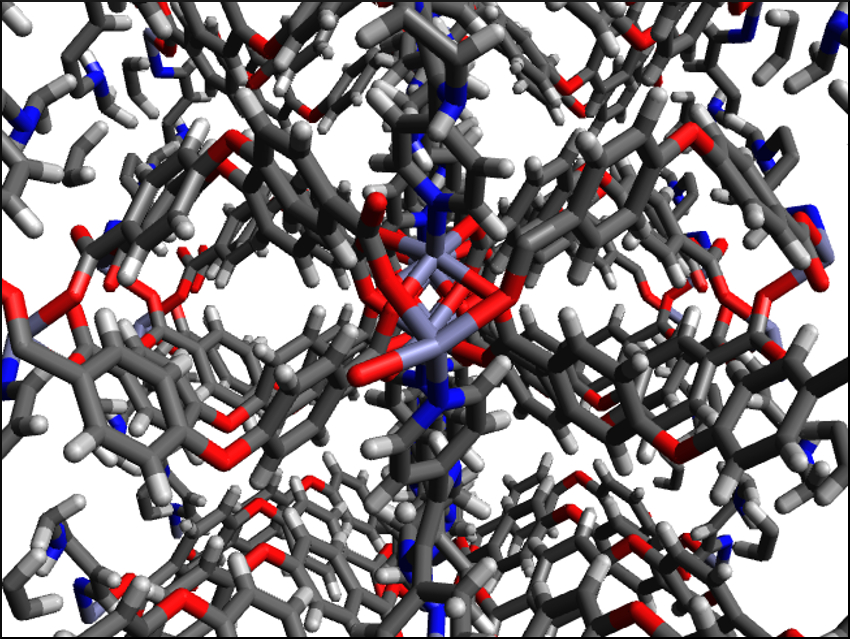
Metal–organic frameworks (MOFs) are porous, crystalline materials with possible applications, e.g., in catalysis or gas separation. They consist of metal ions which are connected by organic ligands. This structure usually leads to a low electric conductivity, which prevents the use of MOFs in electronics.
Ali Morsali, Tarbiat Modares University, Tehran, Iran, and colleagues have developed a MOF (pictured) with tunable electric conductivity. The team first synthesized the MOF Zn(OBA)(L*)]·DMF or TMU-60 (H2OBA = 4,4′-oxybis(benzoic acid), L* = 5,6-di(pyridin-4-yl)-1,2,3,4-tetrahydropyrazine, DMF = dimethylformamide). The framework was prepared from Zn(NO3)2·4H2O, H2OBA, and N,N-bis(pyridin-4-ylmethylene)ethane-1,2-diamine. The diamine ligand was converted to L* in situ. Excess solvent was removed to activate the MOF by washing with CH2Cl2 or by heating the material in a vacuum.
The activated TMU-60 was then shaken in an aqueous solution containing Cd2+ ions. The Cd2+ ions were adsorbed by the framework’s amine groups and the conductivity of the MOF was improved from 5.3·10–5 Scm–1 to 1.8·10–2 Scm–1. According to the researchers, the charge transport is based on a hopping mechanism between the metal nodes and the adsorbed Cd2+ ions. The conductivity can be tuned by varying the amount of Cd2+. These results are promising for the use of MOFs in electronics.
- Highly Electroconductive Metal–Organic Framework: Tunable by Metal Ion Sorption Quantity,
Farzaneh Rouhani, Fatemeh Rafizadeh-Masuleh, Ali Morsali,
J. Am. Chem. Soc. 2019.
https://doi.org/10.1021/jacs.9b04173