C–H activations, usually catalyzed by transition metal complexes, can be used to construct organic molecules. Most often, expensive 4d or 5d transition metals have been used for this type of reaction. Approaches with cheaper, more abundant 3d transition metals, e.g., cobalt, are useful.
Ruhuai Mei, Chengdu University, China, Lutz Ackermann, University of Göttingen, Germany, and colleagues have developed a cobalt-catalyzed, electrochemical C–H/N–H functionalization of benzhydrazides that results in an annulation with 1,3-diynes. The team used the inexpensive Co(OAc)2 as a catalyst, pivalic acid (PivOH) as an additive, and 2,2,2-trifluoroethanol (TFE) as the solvent. The reaction was performed under argon at 60 °C. Electricity was used as a green oxidant, with a glassy carbon anode, a platinum cathode, and a constant current of 4 mA.
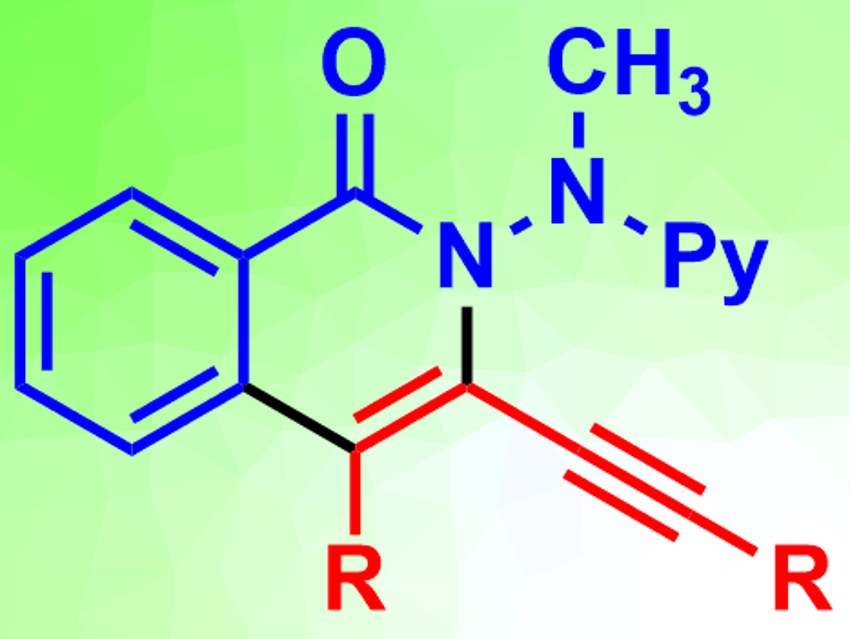
The team reacted both aryl- and alkyl-substituted 1,3-diynes with a range of substituted benzhydrazides. The desired annulation products (pictured) were obtained in moderate to excellent yields. According to the researchers, the use of electricity avoids the need for toxic and/or expensive sacrificial oxidants.
- Cobaltaelectro-Catalyzed Oxidative C–H/N–H Activation with 1,3-Diynes by Electro-Removable Hydrazides,
Ruhuai Mei, Wenbo Ma, Yin Zhang, Xiaoqiang Guo, Lutz Ackermann,
Org. Lett. 2019.
https://doi.org/10.1021/acs.orglett.9b02463