Neptunium is the first transuranium element and has no stable isotopes. For its neighboring element uranium, there are dozens of known peroxide compounds, but much less is known about neptunyl peroxide compounds. H2O2 can act as an oxidizing or reducing agent for neptunium, depending on the conditions.
Peter C. Burns, University of Notre Dame, IN, USA, and colleagues have synthesized the neptunyl peroxide compound Ca2[NpO2(O2)3]·9H2O.. According to the team, it is the first example of an isolated Np(VI) peroxide compound. The team prepared a solution of 237Np(V) in aqueous HCl, which was combined with H2O2, CaCl2, and the base tetramethylammonium hydroxide (TMAOH) to give the desired product as red crystals.
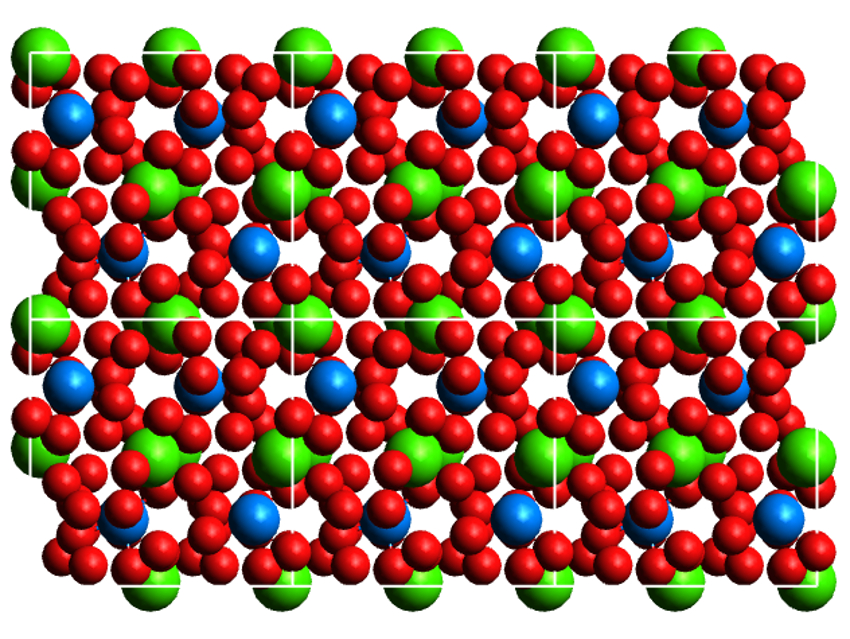
The neptunyl triperoxide product was characterized using X-ray diffraction (structure pictured), Raman spectroscopy, and elemental analysis, among other techniques. It is isostructural to the uranium compound Ca2[UO2(O2)3]·9H2O and has the longest nonbridging Np(VI) neptunyl bonds reported so far. This work could help to better understand the chemistry of neptunium in peroxide-rich conditions.
- Neptunyl Peroxide Chemistry: Synthesis and Spectroscopic Characterization of a Neptunyl Triperoxide Compound, Ca2[NpO2(O2)3]·9H2O,
Sarah Hickam, Debmalya Ray, Jennifer E. S. Szymanowski, Ru-Ye Li, Mateusz Dembowski, Philip Smith, Laura Gagliardi, Peter C. Burns,
Inorg. Chem. 2019.
https://doi.org/10.1021/acs.inorgchem.9b01712