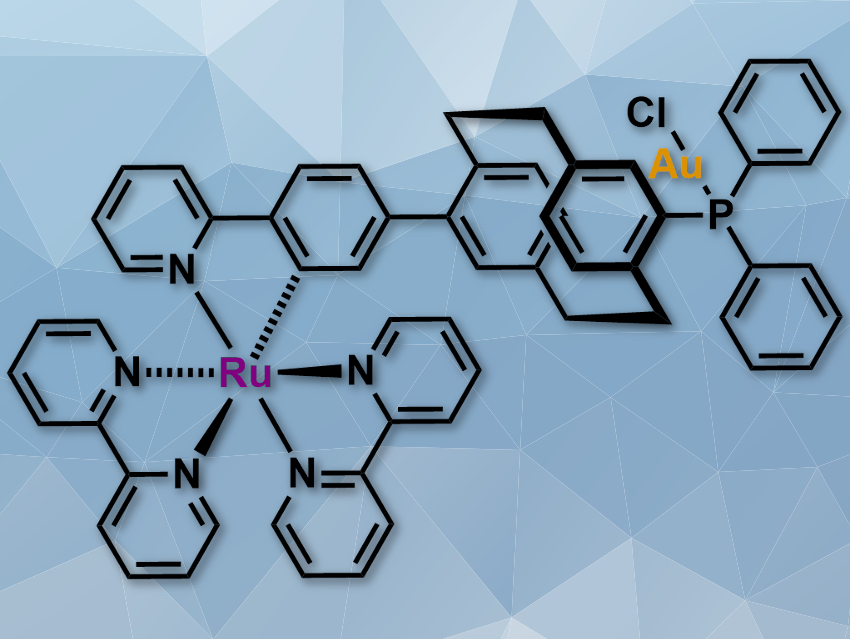
Many transition-metal complexes are useful catalysts. Their properties can be tuned by changing the ligands, which allows for many variations. Combining different metals in one bimetallic complex is another way to change the reactivity of such catalysts. In this case, one metal can enhance the catalytic activity of the other, or the two metals can truly cooperate and both take part in the catalytic process. The latter is rare, with only few known examples for defined binuclear organometallic complexes with cooperative effects.
Stefan Bräse, Karlsruhe Institute of Technology (KIT), Germany, and colleagues have synthesized a gold/ruthenium heterobimetallic complex (pictured) with a [2.2]paracyclophane backbone that ensures a defined distance between the metals. The complex was synthesized from paracyclophane dibromide, which was functionalized with a phosphine and 2-phenylpyridine before introducing the metals and the other ligands.
The team tested the catalytic activity of the complex in a visible-light mediated arylative Meyer-Schuster rearrangement. This reaction combines a propargyl alcohol and an aromatic building block to give an arylated enone. The team’s Au/Ru complex can catalyze this reaction. However, compared with the separate, monometallic complexes, the researchers observed a slight drop in yield. They attribute this to metal-to-metal interactions in the complex. Nevertheless, according to the team, the work could help with the design and the understanding of bimetallic complexes in catalysis applications.
- A Highly Stable, Au/Ru Heterobimetallic Photoredox Catalyst with a [2.2]Paracyclophane Backbone,
Daniel M. Knoll, Christoph Zippel, Zahid Hassan, Martin Nieger, Patrick Weis, Manfred Kappes, Stefan Bräse,
Dalton Trans. 2019.
https://doi.org/10.1039/c9dt04366g