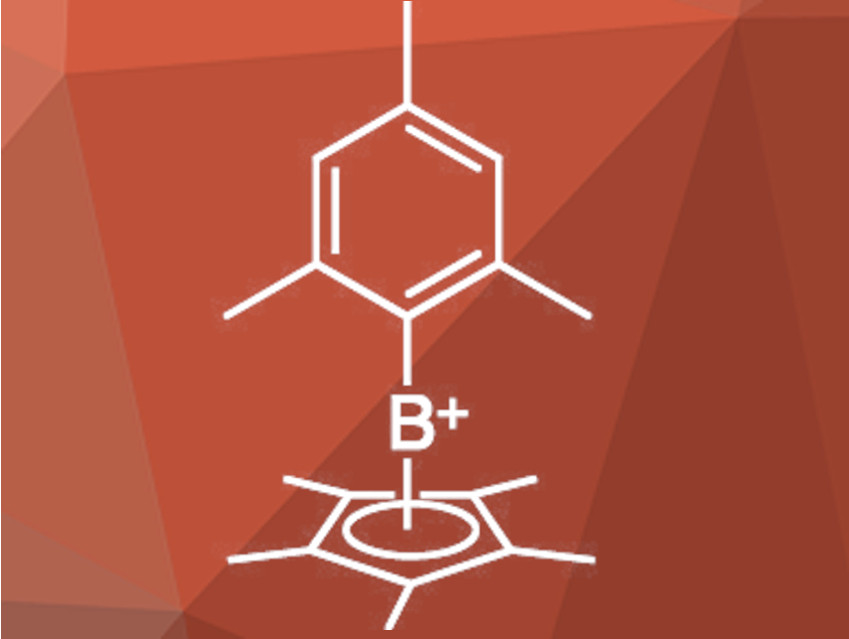
Cationic boron species are useful as catalysts and reagents for organic synthesis. Most boron cations can be placed into one of three distinct groups depending on the coordination number at boron. Dicoordinate borinium cations are highly labile and difficult to isolate due to their strong electron deficiency. Tricoordinate borenium cations can be used as Lewis-acidic catalysts. Tetracoordinate boronium cations are electronically saturated and can undergo reductions or nucleophilic additions.
η5-Pentamethylyclopentadienyl-coordinated boron cations, [η5Cp*B–R]+, combine the properties of boronium and borenium cations. They are electronically saturated but can still act as Lewis acids due to the flexible coordination of Cp*.
Ching-Wen Chiu, National Taiwan University, Taipei, and colleagues have synthesized a [η5Cp*B–R]+ cation with a mesityl group (pictured). The team reacted Cp*BCl2 with mesityllithium in toluene, which gave the chloride salt of the target cation. Anion metathesis with Li[B(C6F5)4] in dichloromethane gave the tetrakis(pentafluorophenyl)borate salt. This product is stable toward air and moisture in solid form. The use of weakly coordinating anions such as B(C6F5)4– is critical because Lewis-basic anions display undesired interactions with the boron cation.
The structure of the new compound was determined using NMR spectroscopy and single-crystal X-ray diffraction. The Lewis-acidic properties were tested by treatment with triethylphospine oxide, which swiftly formed an adduct with the boron cation in dichloromethane. During this reaction, the coordination mode of the Cp* group changes. [η5Cp*B–Mes][B(CF5)4] catalyzes the deoxygenation of aryl ketones in a wet solvent. According to the researchers, the coordinatively flexible substituent at boron is critical for catalytic activity and stability.
- [η5-Cp*B-Mes]+: A Masked Potent Boron Lewis Acid,
Hsi-Ching Tseng, Chao-Tang Shen, Kentaro Matsumoto, Ding-Nan Shih, Yi-Hung Liu, Shie-Ming Peng, Shigehiro Yamaguchi, Ya-Fan Lin, Ching-Wen Chiu,
Organometallics 2019.
https://doi.org/10.1021/acs.organomet.9b00671