TiO2 nanocrystals can be used to store lithium ions, which could make them useful for lithium-ion batteries. However, they have a low electric conductivity, which is a problem when they are used as an electrode in a battery. They also tend to aggregate, which negatively affects their properties. Combining TiO2 nanocrystals with a conductive carbon material could solve these problems. However, existing composite materials are inhomogenous—which reduces their performance—and/or difficult to prepare.
Hai Wang, Guilin University of Technology, China, and colleagues have developed a new TiO2–x@C nanocomposite electrode material for lithium-ion batteries. The team prepared TiO2 nanocrystals from tetrabutyl titanate, using formic acid and anhydrous ethanol to limit crystal growth and sodium dodecylbenzenesulfonate (SDBS) as a surfactant. The resulting nanocrystals were mixed with vitamin C as a carbon source and heated to 200 °C.
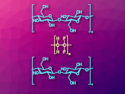
The molten vitamin C and the nanocrystals form a solid-liquid mixture, which ensures a uniform distribution of the TiO2 particles in the composite material (schematically pictured). The vitamin C is converted into a porous, three-dimensional carbon matrix, which is closely connected to the nanocrystals by covalent bonds. The process also introduces surface defects in the TiO2 crystals that improve the material’s performance as an electrode material.
The structure of the nanocomposite combines the good electrical conductivity of the carbon framework with the useful lithium-ion transport and storage properties of TiO2. The carbon matrix also prevents the aggregation of the nanocrystals. The resulting electrodes have high reversible capacities.
- Unusual Improvement of Pseudocapacitance of Nanocomposite Electrodes: Three-Dimensional Amorphous Carbon Frameworks Triggered by TiO2 Nanocrystals,
Huibing Lu, Caihong Yang, Huiming Bao, Linjiang Wang, Cunjun Li, Hai Wang,
ACS Appl. Mater. Interfaces 2019.
https://doi.org/10.1021/acsami.9b17595