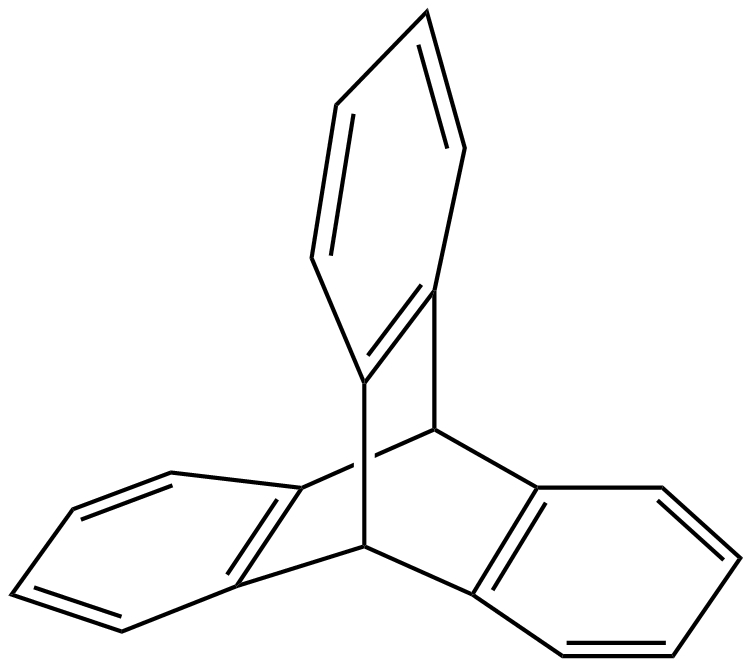
 Iptycenes are molecules composed of arene units bound to a bridged bicyclo-octatriene core. The simplest example of this type of compound is triptycene (pictured right). Building larger iptycene-based structures could lead to useful materials. Carbon nanobelts (CNBs) consist of fused benzene rings that form a belt-like shape. (6,6)CNB, for example, is a cycle composed of 12 benzene rings. The sides of CNBs could be extended via cycloaddition reactions to give iptycenes.
Iptycenes are molecules composed of arene units bound to a bridged bicyclo-octatriene core. The simplest example of this type of compound is triptycene (pictured right). Building larger iptycene-based structures could lead to useful materials. Carbon nanobelts (CNBs) consist of fused benzene rings that form a belt-like shape. (6,6)CNB, for example, is a cycle composed of 12 benzene rings. The sides of CNBs could be extended via cycloaddition reactions to give iptycenes.
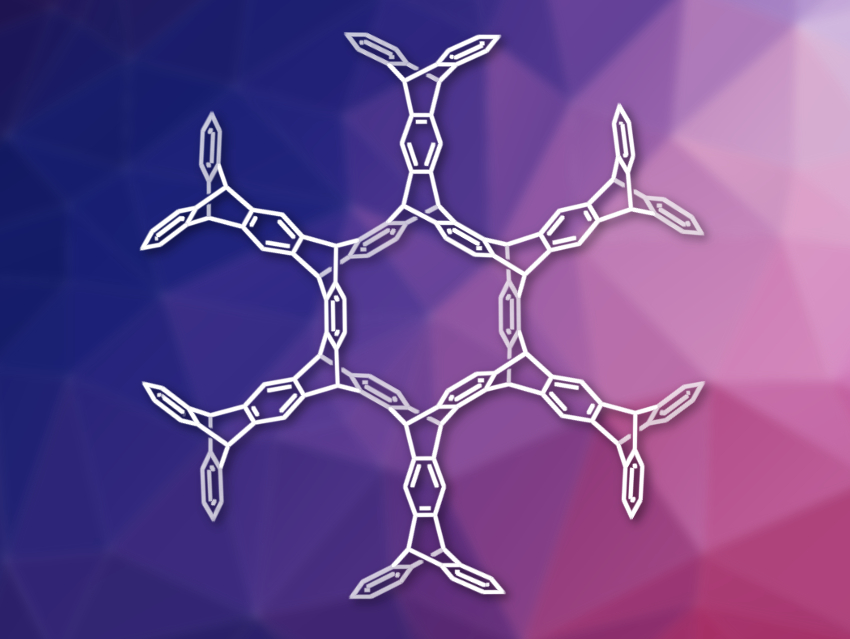
Yasutomo Segawa, Nagoya University, Japan, Institute for Molecular Science and Graduate University for Advanced Studies (SOKENDAI), both Okazaki, Japan, Kenichiro Itami, Nagoya University, and colleagues have synthesized the largest iptycene to date using carbon nanobelts as a starting material. The team reacted (6,6)CNB with arynes or alkynes in a six-fold Diels–Alder reaction to extend the carbon nanobelt outwards. To obtain a large cycloiptycene, they used a triptycene derivative to extend the CNB. The researchers used a triptycene substituted with neighboring SiMe3– and triflate groups as an aryne precursor and reacted it with (6,6)CNB in the presence of CsF in a mixture of acetonitrile and 1,2-dicholorethane at 85 °C for 7 d.
The desired product, tetracosipticene (pictured), was obtained in an isolated yield of 5 %. According to the team, it is the largest iptycene reported to date. The researchers performed density functional theory (DFT) calculations and found that the release of strain is a driving force behind the reaction. The work shows the utility of CNBs as building blocks for new carbon-based nanostructures.
- Synthesis of Cycloiptycenes from Carbon Nanobelt,
Hiroki Shudo, Motonobu Kuwayama, Yasutomo Segawa, Kenichiro Itami,
Chem. Sci. 2020.
https://doi.org/10.1039/d0sc02501a