Some transition-metal complexes, such as cisplatin (cis-[PtCl2(NH3)2]), can be used as anticancer drugs. Many other metal complexes have been tested for their biological activity. However, finding drug candidates that have few side effects and target cancer cells specifically is still challenging. Thiosemicarbazones (R2N–C(=S)–NR–N=CR2) are ligands with a range of biological activities, including anticancer effects.
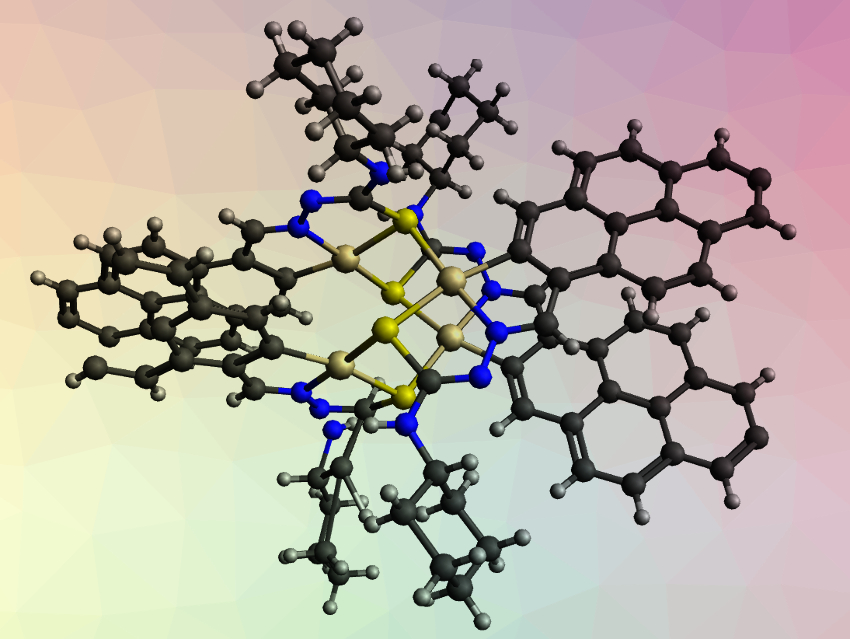
Victor M. Deflon, University of São Paulo, São Carlos, Brazil, Peter J. Sadler, University of Warwick, UK, and colleagues have used thiosemicarbazone ligands with a pyrene unit to synthesize tetranuclear palladium(II) and platinum(II) complexes with potential anticancer activity. The team obtained complexes of the type [M4(μ-S-PrR-κ3–C,N,S)4] (M = Pd, Pt; PrR = pyrene-derived thiosemicarbazone ligand with substitute R; R = ethyl, cyclohexyl; example with cyclohexyl groups pictured) from trans-[PdCl2(MeCN)2] or K2[PtCl4] and the respective ligand. The ligands bind to the metal atoms via the thiosemicarbazone’s sulfur atom, the imine-type nitrogen atom, and a carbon atom of the pyrene unit.
The team used X-ray crystallography to investigate the structure of the cyclohexyl-substituted Pt complex. They found that the complex consists of four sulfur-bridged monometallic units, forming an eight-membered ring of alternating Pt and sulfur atoms (pictured in yellow/gold). The team found that the ethyl-substituted complexes inhibit the growth of an ovarian cancer cell line. The cytotoxic activity of the Pd complex is comparable to cisplatin, while the Pt complex shows a much higher activity. The two complexes also have low toxicity towards a non-cancerous human cell line, showing their selectivity for cancer cells.
- Novel tetranuclear PdII and PtII anticancer complexes derived from pyrene thiosemicarbazones,
Carolina G. Oliveira, Isolda Romero-Canelón, James P. C. Coverdale, Pedro Ivo S. Maia, Guy J. Clarkson, Victor M. Deflon, Peter J. Sadler,
Dalton Trans. 2020.
https://doi.org/10.1039/d0dt01133a