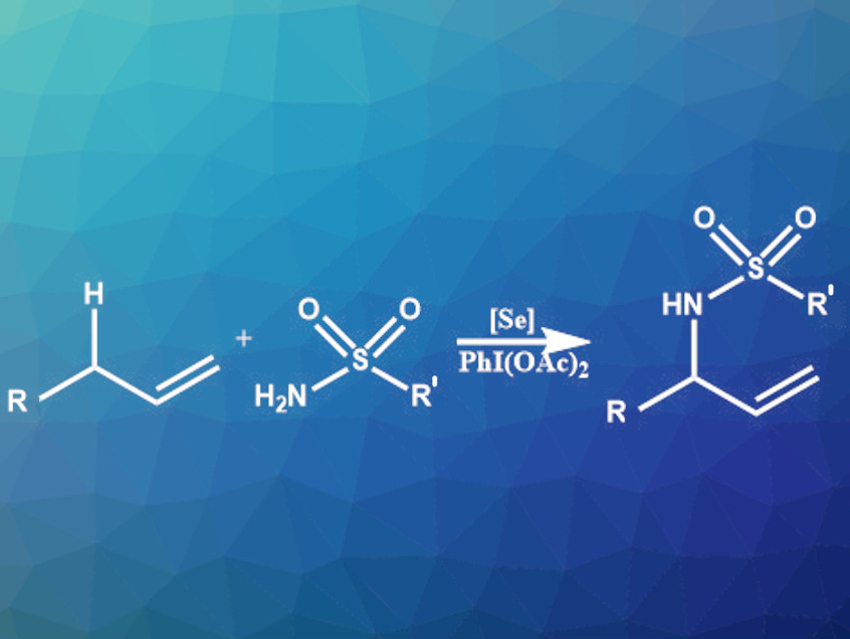
Terpenes and terpenoids are structurally diverse natural products made from isoprene-like precursors. C–H aminations at the allylic position of these precursors could allow the preparation of new nitrogen-containing, potentially bioactive compounds. However, it can be challenging to achieve regioselectivity in this type of reaction. Existing site-selective approaches for the allylic C–H amination of alkenes are hampered by limited yields, hazardous nitrogen sources, and a need for stoichiometric amounts of selenium reagents.
Forrest E. Michael and colleagues, University of Washington, Seattle, USA, have developed a broadly applicable, metal-free allylic C–H amination reaction that requires only catalytic amounts of selenium and uses easily accessible sulfonamide or sulfamate coupling partners (pictured). The team used tricyclohexylphosphine selenide (Cy3PSe) as the catalyst and PhI(OAc)2 as an oxidizing agent. The reaction was performed at room temperature to 40 °C in dichloromethane (DCM). It gives moderate to excellent yields and provides high regioselectivity. The conversion of highly substituted alkenes using Cy3PSe was not successful. However, the team found that using an alternative N-heterocyclic carbene-derived selenourea catalyst provided high yields for these substrates.
The process can be used for the functionalization of a wide range of terpenoid natural products, such as citronellol, geraniol, or carvone. In addition, sulfonamides and sulfamates derived from biologically active alcohols, such as the antifungal paclobutrazol or the antibiotic triclosan, reacted readily with different alkenes, leading to drug conjugates. Overall, the method is useful for the synthesis of new classes of potentially bioactive products.
- Catalytic Metal-free Allylic C–H Amination of Terpenoids,
Wei Pin Teh, Derek C. Obenschain, Blaise M. Black, Forrest E. Michael,
J. Am. Chem. Soc. 2020.
https://doi.org/10.1021/jacs.0c06997