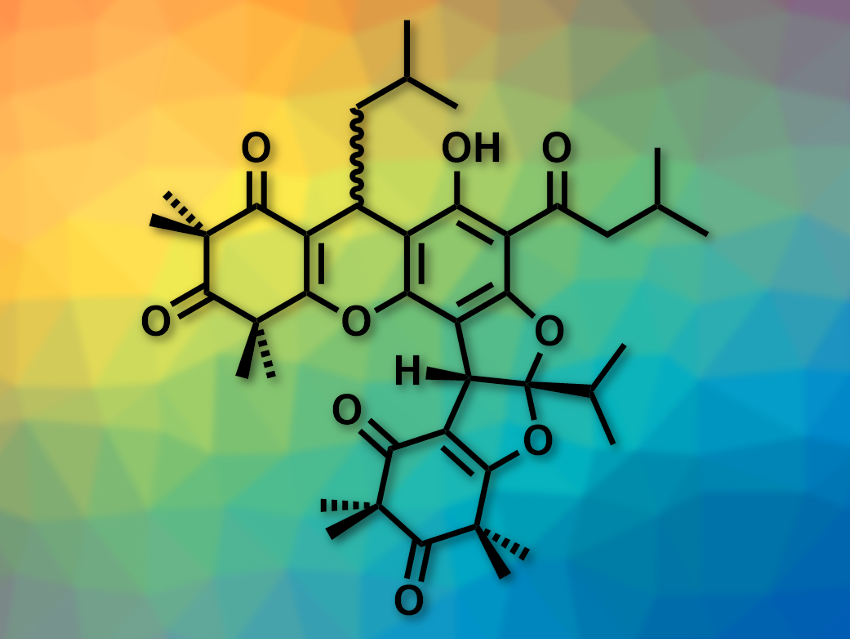
Polycyclic polymethylated phloroglucinols (PPPs) are plant metabolites most often found in plants of the myrtle family or Guttiferae plants. PPPs have interesting, complex structures and can have useful biological activities, e.g., antibacterial, anti-inflammatory, or antiplasmodial effects. Antiplasmodial compounds fight parasites like those that cause malaria.
Tomentosones A and B (pictured), for example, are PPPs that were isolated from the rose myrtle shrub. These compounds have a 6/6/6/5/5/6 fused-ring system and differ only in the orientation of one isobutyl side chain. Tomentosone A has good antimalarial activity, while tomentosone B is significantly less active.
Yingjie Hu, Guangzhou University of Chinese Medicine, China, Hongxin Liu, Guangdong Provincial Key Laboratory of Applied Botany, Chinese Academy of Sciences, and Guangdong Academy of Sciences, China, Haibo Tan, Guangdong Provincial Key Laboratory of Applied Botany and Central South University, Changsha, China, and colleagues have performed the first biomimetic total syntheses of tomentosones A and B. The team based their syntheses on a possible biosynthetic pathway for tomentosones A and B.
The researchers first prepared an α,β-unsaturated ketone intermediate and reacted it with phloroglucinol (a benzenetriol) via a Michael addition. This was followed by an intramolecular dehydration and a Friedel–Crafts acylation to give rhodomyrtone, which contains the targeted 6/6/6 ring system. Rhodomyrtone was then subjected to a dehydroxylation/Michael addition/Kornblum–DeLaMare peroxide rearrangement cascade reaction with a peroxide intermediate to introduce the remaining rings and give tomentosones A and B. According to the researchers, this work could be useful for biosynthesis investigations, structure–activity relationship studies, and drug development involving these natural products.
- The Biomimetic Total Syntheses of the Antiplasmodial Tomentosones A and B,
Xiao Zhang, Chunmao Dong, Guiyun Wu, Luqiong Huo, Yunfei Yuan, Yingjie Hu, Hongxin Liu, Haibo Tan,
Org. Lett. 2020.
https://doi.org/10.1021/acs.orglett.0c02943