Disulfide bridges between cysteine side chains are important connections in peptide and protein chemistry. For the controlled, sequential formation of multiple disulfide bridges, protecting groups are needed. Disulfide protecting groups, which can be removed by disulfide reducing agents, are useful for the start of such a sequence. However, examples of this type of protecting group are limited to -StBu and S-2,6-dimethoxybenzyl (S-DMP). These groups have drawbacks, such as inefficient deprotection or incompatibility with solid-phase peptide synthesis (SPPS).
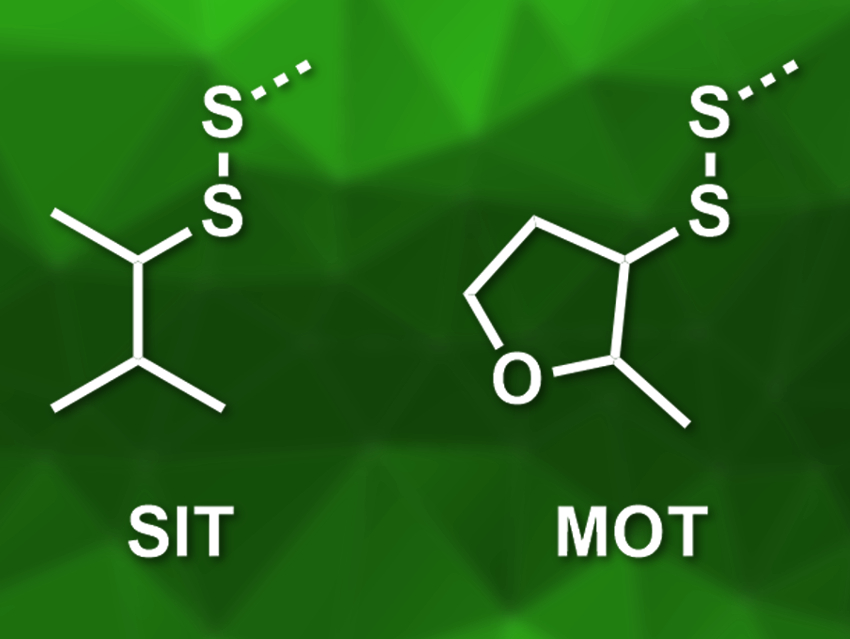
Fernando Albericio, University of KwaZulu-Natal, Durban, South Africa, Institute for Advanced Chemistry of Catalonia, Barcelona, and University of Barcelona, Spain, Beatriz G. de la Torre, University of KwaZulu-Natal, and colleagues have developed two new disulfide-based protecting groups, sec-isoamyl mercaptan (SIT) and 2-methyloxolane-3-thiol (MOT) (both pictured in disulfide form). Both of these thiols are commercially available, and SIT- or MOT-protected cysteine building blocks for peptide synthesis can be prepared in good yields.
The SIT and MOT protecting groups are based on secondary thiols with a β-methyl substituent. This provides the right level of steric bulk to ensure they are easy to remove, but stable enough for use in solid-phase peptide synthesis. Both groups can be reliably removed with the reducing agent 1,4-dithiothreitol (DTT). The SIT group showed particularly good performance in SPPS, and thus, could be a good candidate for cysteine side-chain protection according to the researchers.
- Disulfide-Based Protecting Groups for the Cysteine Side Chain,
Amit Chakraborty, Anamika Sharma, Fernando Albericio, Beatriz G. de la Torre,
Org. Lett. 2020.
https://doi.org/10.1021/acs.orglett.0c03705