Malaria is a mosquito-borne disease that is widespread in the tropical and subtropical regions of the world. Multidrug-resistant strains of the Plasmodium parasite that causes malaria have led to a need for new treatment options. Biologically active natural products can be useful starting points for the search for new drug candidates. The Australian marine sponge Citronia astra, for example, has been a source for several biologically active compounds.
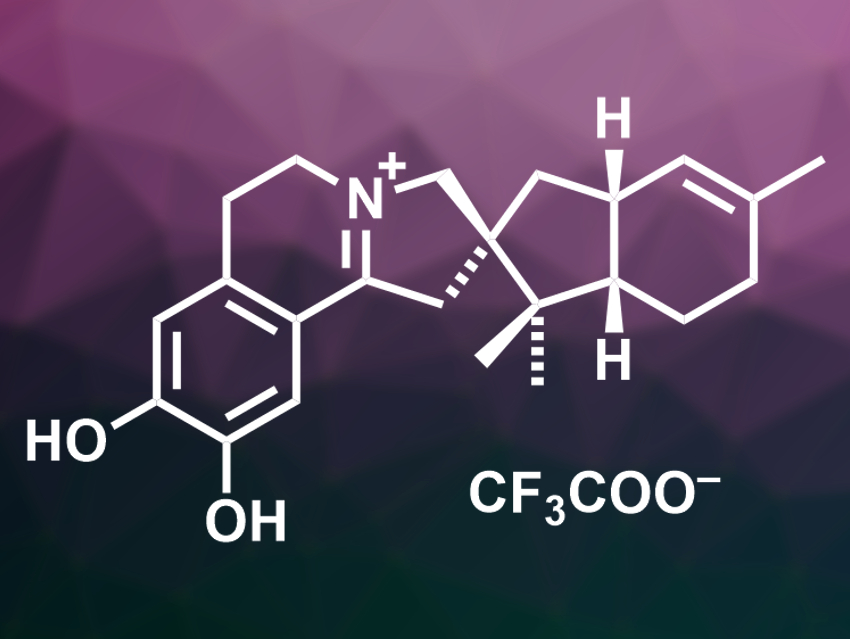
Anthony R. Carroll, Griffith University, Southport, Australia, and colleagues have isolated citronamine A (pictured), an isoquinoline alkaloid, from Citronia astra, determined its structure, and investigated its activity against the malaria-causing parasite. The team extracted 10 kg of the marine sponge with water and used column chromatography to separate the compounds in he crude extract. They obtained citronamine A in the form of its trifluoroacetate.
The researchers characterized the compound using high-resolution electrospray-ionization mass spectrometry (HRESIMS), 1H and 13C NMR spectroscopy, and electronic circular dichroism (ECD) spectroscopy. This allowed them to determine the structure of citronamine A, which features an unusual pentacyclic system and an isoquinoline-type unit. Isoquinoline derivatives can have antiplasmodial properties. Therefore, the team tested the activity of citronamine A against different strains of Plasmodium parasites. It showed moderate activity against both drug-sensitive and drug-resistant strains.
- Citronamine A, an Antiplasmodial Isoquinoline Alkaloid from the Australian Marine Sponge Citronia astra,
Dale W. Prebble, Darren C. Holland, Luke P. Robertson, Vicky M. Avery, Anthony R. Carroll,
Org. Lett. 2020.
https://doi.org/10.1021/acs.orglett.0c03633