Renewable energy sources such as wind and solar power could help decrease the need for fossil fuels. However, this requires a safe, cost-effective way to store the energy due to the fluctuating nature of these sources. Lithium-ion batteries could be used for this, but they suffer from safety issues and the limited availability of lithium. Aqueous zinc-based batteries could be useful alternatives for grid-scale energy storage. They are safer, have a high energy density, and the materials used to make them are abundant. However, the rechargeable zinc batteries developed so far have thick zinc metal anodes, which contain a large excess of zinc that increases cost. Also, the anodes are prone to forming dendrites, which can short-circuit the battery.
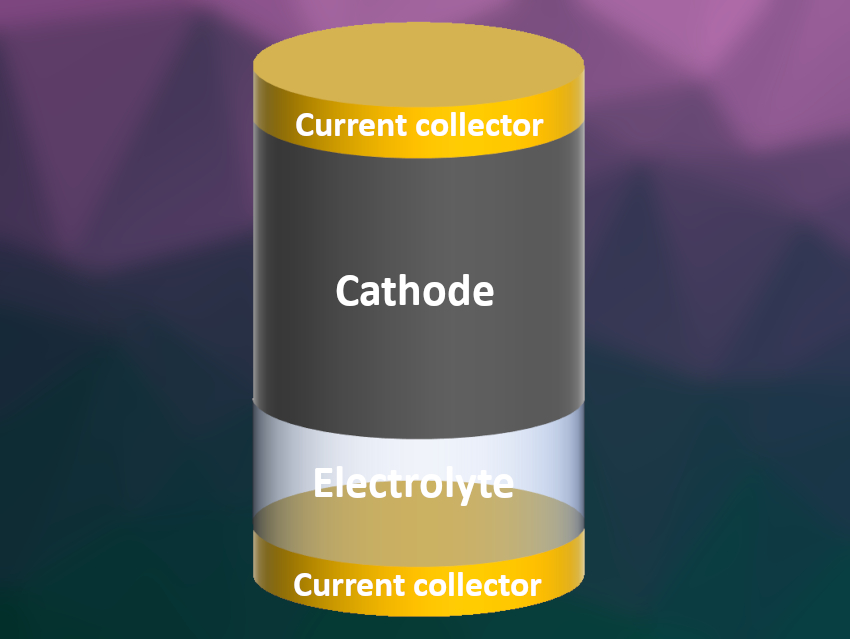
Yunpei Zhu, Husam N. Alshareef, King Abdullah University of Science and Technology (KAUST), Thuwal, Saudi Arabia, Yi Cui, Stanford University, CA, USA, and SLAC National Accelerator Laboratory, Menlo Park, CA, USA, have developed an anode-free design for a Zn–MnO2 battery. In this battery, a zinc-rich cathode is the sole source for zinc plating onto a copper current collector (schematically pictured). The team used a manganese dioxide cathode that they pre-intercalated with zinc ions, an aqueous zinc trifluoromethanesulfonate electrolyte solution, and copper foil current collectors.
During charging, zinc metal gets plated onto the copper foil, and during the discharge process, the metal is stripped off. The researchers coated the copper current collector with a layer of carbon nanodiscs. This layer promoted uniform zinc plating, prevented dendrites, and increased the efficiency of zinc plating and stripping. The battery showed high efficiency, energy density, and stability, retaining 62.8 % of its storage capacity after 80 charging and discharging cycles.
- An Anode-Free Zn–MnO2 Battery,
Yunpei Zhu, Yi Cui, Husam N. Alshareef,
Nano Lett. 2021.
https://doi.org/10.1021/acs.nanolett.0c04519