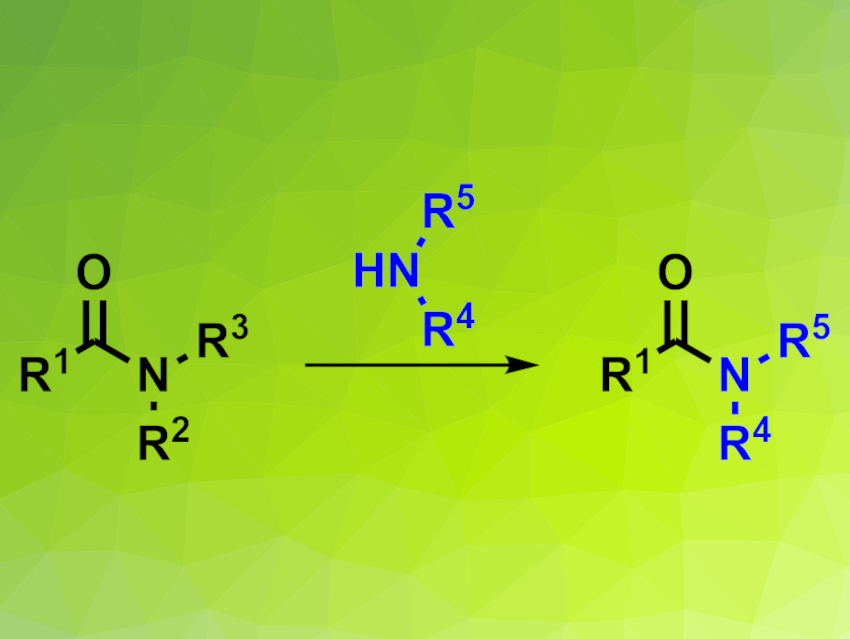
Amides are an important class of organic molecules. Many natural products, pharmaceuticals, and agrochemicals contain amide groups, and amides also serve as important intermediates in organic synthesis. Amides are usually synthesized by the amidation of carboxylic acids or via transamidation reactions. The latter is better suited for the synthesis of highly functionalized amides because it can often be carried out under milder conditions. Transamidations offer a convenient way to convert easily synthesizable amides to biologically more active species. The transamidation of primary and secondary amides is usually straightforward. In contrast, transamidation reactions of tertiary alkyl amides are more challenging.
Jun-An Ma, Chi Wai Cheung, Tianjin University, China, and colleagues have developed a broadly applicable tungsten-catalyzed transamidation process for tertiary alkyl amides (pictured). The amide starting material was reacted with an amine in the presence of WCl6 as a catalyst, 1,10-phenanthroline as a ligand, and chlorotrimethylsilane as a Lewis base that activates the amide bond. The reaction was carried out in N-methylpyrrolidone (NMP) at 140 °C over 36 h.
The team obtained the desired amides in moderate to excellent yields, using a wide range of aromatic, aliphatic, and heterocyclic amines carrying both electron-donating and electron-withdrawing groups. The scope of amide starting materials is also broad, allowing for both aromatic and aliphatic substituents at the amino units as well as a wide variety of aliphatic side chains. The researchers used their approach to synthesize different biologically active molecules, such as derivatives of the antidepressant citalopram and the sedative zolpidem.
- Tungsten-Catalyzed Transamidation of Tertiary Alkyl Amides,
Fang-Fang Feng, Xuan-Yu Liu, Chi Wai Cheung, Jun-An Ma,
ACS Catal. 2021.
https://doi.org/10.1021/acscatal.1c01840