Evonik is working with Stanford University on a technology to deliver mRNA to tissues and organs that goes beyond the capabilities of lipid nanoparticles (LNPs). This polymer-based platform complements Evonik’s existing portfolio of lipid-based drug delivery, including LNPs. Starting this month, Evonik and Stanford scientists will begin a three-year sponsored research collaboration to develop the polymer-based drug delivery system, which Evonik will license and commercialize.
Effective and safe delivery of mRNA into cells is one of the biggest challenges for expanding the use of mRNA therapeutics to promising fields such as cancer immunotherapy, protein replacement, and gene editing.
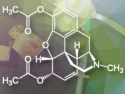
Robert Waymouth, Paul Wender, and Ronald Levy of Stanford University have developed a new polymer-based delivery platform known as CART (Charge Altering Releasable Transporters). The Evonik and Stanford University scientists want to scale up the synthesis and formulation, and further develop this innovative technology for organ selective delivery based on a non-animal-derived, synthetic degradable polymer. As one of the few integrated development and manufacturing partners for gene therapies, Evonik aims to make this technology available in GMP quality (Good Manufacturing Practice) for use in clinical-stage developments and ultimately on a commercial scale.
Evonik recognized the potential of gene-based therapeutic approaches at an early stage. In 2016, they bought Transferra Nanosciences, Vancover, Canada, which has a strong focus on developing parenteral drug formulations using lipid nanoparticles and liposomes, and in 2020, Wilshire Technologies, Princeton, NJ, USA, which among other things produces PhytoChol®, a non-animal-derived cholesterol used in many commercial parenteral pharmaceutical products.
- Evonik AG, Essen, Germany
- Stanford University, CA, USA