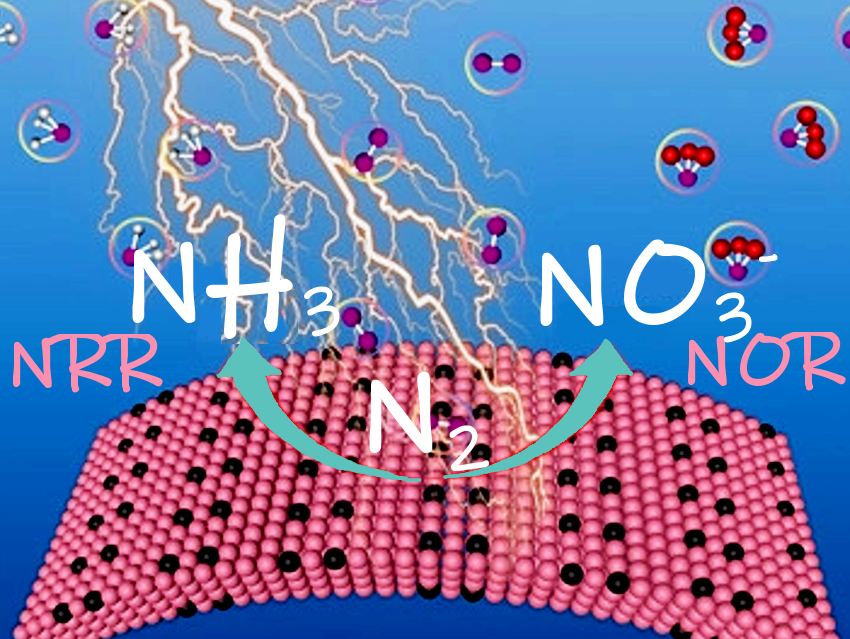
Electrochemical nitrogen fixation under mild conditions could save energy and greenhouse gas emissions in industrial nitrogen production. Recently, many pioneering catalysts have been reported. However, a multifunctional N2 reduction reactions (NRR)/N2 oxidation reaction (NOR) catalyst with high ammonia yield, large Faradaic efficiency, and excellent stability, which effectively suppresses the side reactions during N2 fixation, is still missing.
Yongwen Tan, Hunan University, China, and colleagues have designed a nanoporous boron carbide (np-B13C2) catalyst for electrochemical nitrogen fixation. The team used a chemical etching approach to dissolve an orthorhombic CoB solid solution phase of a Co30B64C6 precursor. What remains is a B13C2 skeleton with a bicontinuous nanoporous structure. The researchers found that this np-B13C2 shows superior catalytic activity and stability to NRR and NOR in aqueous electrolytes compared to most reported electrocatalysts.
Strong coupling between the B–C sites affects the electronic structures of the neighboring B atoms of B13C2. As a result, the B sites can effectively adsorb and activate chemically inert N2 molecules. This minimizes the energy required by the potential-determining step. In addition, the introduction of carbon can increase the inherent conductivity and decrease the binding energy of the reactants. As a result, the N2 fixation performance is improved.
A high NH3 yield of 91.28 µg h−1mgcat.−1 and Faradaic efficiency (FE) of 35.53 % at −0.05 V versus the reversible hydrogen electrode were obtained for NRR, as well as long-term stability of up to 70 h. According to the researchers, this means it is among the most active NRR electrocatalysts. The catalyst can also achieve a NO3− yield of 165.8 µg h−1mgcat.−1 and a FE of 8.4 % for NOR.
- Nanoporous B13C2 towards Highly Efficient Electrochemical Nitrogen Fixation,
Jiao Lan, Min Luo, Jiuhui Han, Ming Peng, Huigao Duan, Yongwen Tan,
Small 2021.
https://doi.org/10.1002/smll.202102814