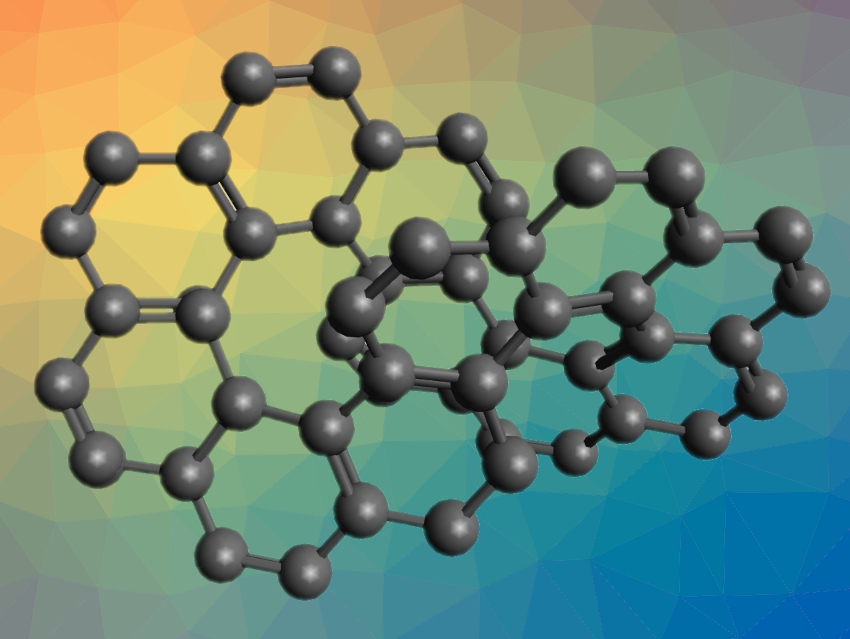
Polycyclic aromatic hydrocarbons (PAHs) can form a variety of interesting shapes. They can, for example, contain loops, such as in circulenes or carbon nanobelts. Open structures can form a “twist”, such as in helicenes. Combining these features could lead to a twisted loop—with a shape similar to an infinity sign.
Maciej Krzeszewski, Hideto Ito, and Kenichiro Itami, Nagoya University, Japan, have synthesized such a molecule, namely, cyclo[c.c.c.c.c.c.e.e.e.e.e.e]dodecakisbenzene (pictured) and called it infinitene. The compound consists of twelve consecutively fused benzene rings forming a twisted loop. The team first prepared a dithiacyclophane macrocycle and performed a Stevens rearrangement to shorten the bridges between the two aromatic fragments. The resulting intermediate was selectively oxidized at the two sulfur atoms remaining on the bridges. Then it was pyrolyzed to remove the sulfur-containing groups and obtain a cyclophadiene. A final photocyclization step gave infinitene as a bench-stable yellow solid.
The product is soluble in organic solvents (unlike, e.g., kekulene), which the team attributes to its nonplanar structure. Its structure was confirmed using X-ray crystallography. Infinitene is chiral, and the racemic mixture obtained can be separated using chiral high-performance liquid chromatography (HPLC).
- Infinitene: A Helically Twisted Figure-Eight [12]Circulene Topoisomer,
Kenichiro Itami, Maciej Krzeszewski, Hideto Ito,
ChemRxiv 2021.
https://doi.org/10.33774/chemrxiv-2021-pcwccThe research has been published as a preprint and has not yet been peer-reviewed.




This research has been published in a peer-reviewed journal, see https://pubs.acs.org/doi/10.1021/jacs.1c10807