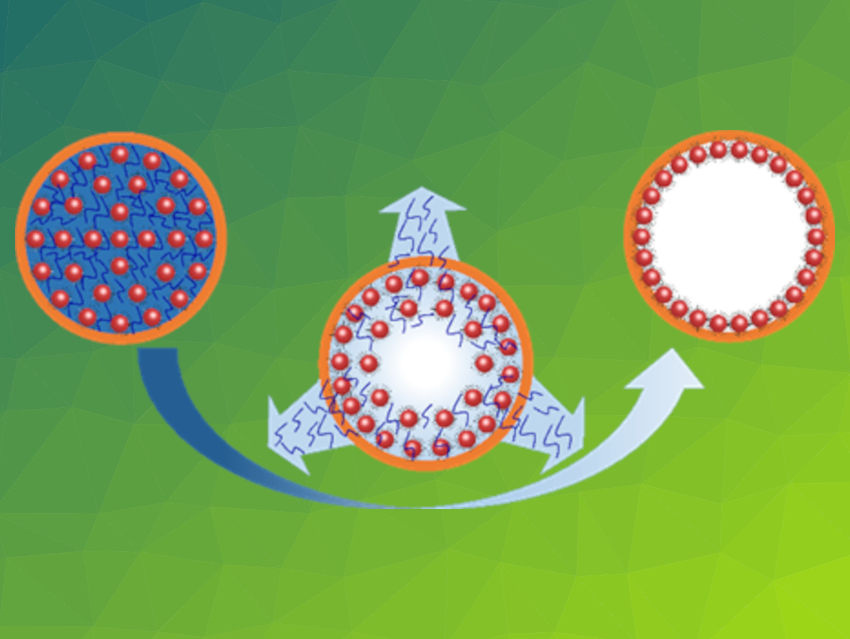
Hollow superstructures of inorganic nanoparticles have attracted research attention due to their controllable permeability, convenient functionalization, and efficient surface utilization. Usually, they are produced by assembling nanoparticles using expensive sacrificial templates. This limits their potential applications.
Yadong Yin, University of California, Riverside, USA, and colleagues have developed a general strategy for the fabrication of submicron hollow superstructures (pictured schematically. The team first co-assembled colloidal nanoparticles (γ-Fe2O3) with organic additives (oleic acid and its oligomers) to form clusters, then coated the clusters with a polymer shell (resorcinol formaldehyde), and finally removed the organic additives and re-dispersed the nanoparticles by exposing them to a good solvent. The key to this process is the re-assembly of nanoparticles against the polymer shells driven by the capillary force during solvent evaporation (pictured in the center), producing hollow superstructures (pictured on the right).
The assembly process can be controlled by the choice of solvents and their evaporation rates. The shell thickness of the nanoparticle superstructures can also be controlled by changing the total amount of organic additives, as this value directly determines the free space inside the polymer shells.
After re-assembling the nanoparticles into hollow superstructures, the polymer shell can be converted into carbon by calcination while retaining the morphology of the hollow superstructures. The conversion of the shell into carbon may also provide opportunities for broader applications. The assembly method can also be used with other inorganic nanoparticles and different additives. Overall, the work provides a general strategy for assembling colloidal nanoparticles into hollow superstructures.
- Self‐assembly of colloidal nanoparticles into encapsulated hollow superstructures,
Chaolumen Wu, Zhiwei Li, Yaocai Bai, Dung To, Nosang V. Myung, Yadong Yin,
Aggregate 2021.
https://doi.org/10.1002/agt2.146