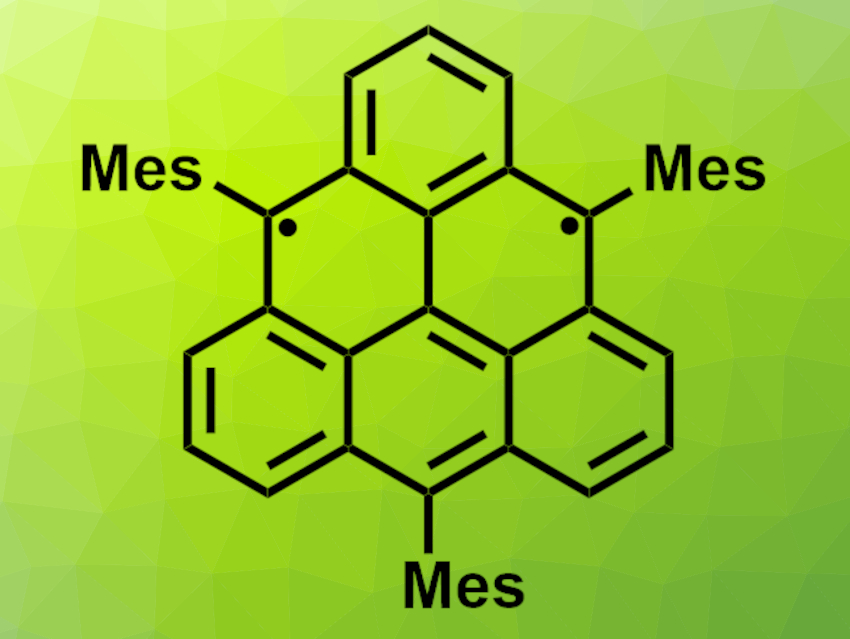
Triangulene, also known as Clar’s hydrocarbon, is a diradical and consists of six benzenoid rings that form a triangular shape. Open-shell graphene fragments such as triangulene could have applications, e.g., in spintronics (electronics that depend not only on charge, but also on electron spin). Triangulene has a triplet ground state and is highly reactive. This reactivity makes triangulene difficult to synthesize. However, bulky substituents can be used to prepare triangulene derivatives with higher stability.
Tomáš Šolomek, University of Bern, Switzerland, and Prievidza Chemical Society, Slovak Republic, Sabine Richert, University of Freiburg, Germany, Michal Juríček, University of Zurich, Switzerland, and Prievidza Chemical Society, and colleagues have synthesized trimesityltriangulene (pictured, Mes = mesityl), a persistent triangulene derivative. The team started from anthrone, which was converted to 9-bromo-10-mesitylanthracene and coupled to (2,6-bis(methoxymethyl)phenyl)-boronic acid. The resulting intermediate has four of the desired six benzenoid rings. It was converted to a dialdehyde in three steps, which then underwent a cyclization in the presence of mesitylmagnesium bromide to close the last two rings. The resulting dihydro-triangulenes were oxidized to obtain the desired triangulene derivative.
The team found that trimesityltriangulene has a triplet character, based on electron paramagnetic resonance (EPR) spectroscopy and density functional theory (DFT) calculations. According to the researchers, the bulky mesityl substituents sterically shield all reactive sites of the product, which kinetically stabilizes the diradical. The product is stable for several weeks in solution at room temperature. The work could inspire the synthesis of further triangulene analogs that might be useful in spintronic materials.
- Trimesityltriangulene: A Persistent Derivative of Clar’s Hydrocarbon,
Leoš Valenta, Maximilian Mayländer, Pia Kappeler, Olivier Blacque, Tomas Solomek, Sabine Richert, Michal Juríček,
Chem. Commun. 2022.
https://doi.org/10.1039/d2cc00352j