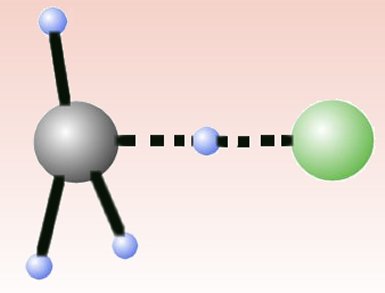
In 1986, John Polanyi shared the Nobel Prize in chemistry for developing general rules for how different forms of energy affect the rates of reactions. The rules state that vibrational energy is more efficient than translational energy in activating a late-barrier reaction, whereas the reverse is true for an early-barrier reaction. Recently, molecular beam experiments by Kopin Liu and co-workers, Institute of Atomic and Molecular Sciences, Taiwan, showed that the late-high-barrier Cl + CHD3 reaction does not conform to the rules.
Gábor Czakó and Joel Bowman, Emory University, Atlanta, USA, have taken a theoretical approach to this anomalous reaction. They have determined the global potential energy surface (PES) of the reaction and performed dynamics calculations with the PES. They confirm that, in contrast to the Polanyi rules, translational energy is just as effective in promoting the reaction as vibrational excitation at low collision energies. They attribute this to long-range steric forces — which pre-align the reactants — playing a larger role than the Polanyi rules consider.
- Dynamics of the Reaction of Methane with Chlorine Atom on an Accurate Potential Energy Surface
G. Czakó, J. M. Bowman,
Science 2011, 334(6054), 343–346.
DOI: 10.1126/science.1208514