Researchers at GlaxoSmithkline (GSK) in Madrid, Spain, and their colleagues have identified 47 quality starting points for pharmaceutical lead optimization for new antimalarial drugs. The team searched the GSK database of 13,533 chemical structures published in 2010.
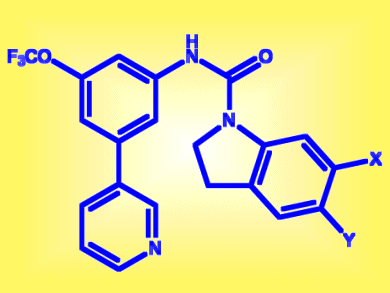
One of the most promising compounds was TCMDC-139046, which has an indoline core, and is known to have anxiolytic activity through its interaction with serotonin antagonist receptors 5-HT2. If it were to be used as an antimalarial, it would have side-effects because of this activity. As such, the GSK team working with the Medicines for Malaria Venture (MMV), Geneva, Switzerland, have used a double-divergent structure–activity relationship (SAR) study to home in on a chemical cousin that lacks the receptor activity but works as an antimalarial. They found better selectivity was obtained with the piperidine derivatives (pictured), where no interaction with 5-HT2c was detected and the activity against multidrug resistant malarial strains was maintained.
- A Divergent SAR Study Allows Optimization of a Potent 5-HT2c Inhibitor to a Promising Antimalarial Scaffold,
F. Calderón, J. Vidal-Mas, J. Burrows, J. C. de la Rosa, M. Belén Jiménez-Díaz, T. Mulet, S. Prats, J. Solana, M. Witty, F. J. Gamo, E. Fernández,
ACS Med. Chem. Lett. 2012.
DOI: 10.1021/ml300008j