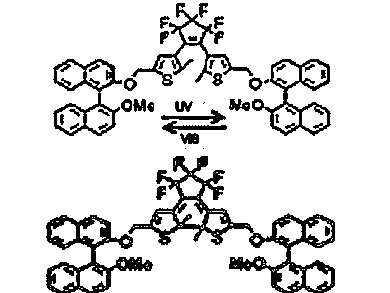
Chirality switching is attractive for the dynamic control of the electronic and optical properties in nanoscale materials. The ability to photochemically switch the chirality in liquid crystals (LCs) is especially intriguing given their potential applications in electro-optic displays, optical data storage, and the asymmetric synthesis of organic molecules and polymers. Kazuo Akagi, Kyoto University, Japan, and colleagues present a dynamic photoswitching of the helical inversion in chiral nematic LCs (N*-LCs) that contain photoresponsive axially chiral dopants.
Photoresponsive chiral dithienylethene derivatives bearing two axially chiral binaphthyl moieties were synthesized. The dihedral angle of the binaphthyl rings changes via the photoisomerization between the open and closed forms of the dithienylethene moiety. The photoresponsive binaphthyl derivatives were added as chiral dopants into N-LCs composed of phenylcyclohexyl or cyanobiphenyl mesogen, which induced N*-LCs.
These compounds are the first chiral dopants that induce a helical inversion in N*-LC via the photoisomerization between open and closed forms of the dithienylethene moiety. The photoresponsive LCs should be applicable for advanced optical memory devices with multiple read−write functions and for helical sense-controllable asymmetric reaction fields.
- Dynamic Photoswitching of Helical Inversion in Liquid Crystals Containing Photoresponsive Axially Chiral Dopants,
Hiroyuki Hayasaka, Tatsuaki Miyashita, Masaru Nakayama, Kenji Kuwada, Kazuo Akagi,
J. Am. Chem. Soc. 2012.
DOI: 10.1021/ja2088053