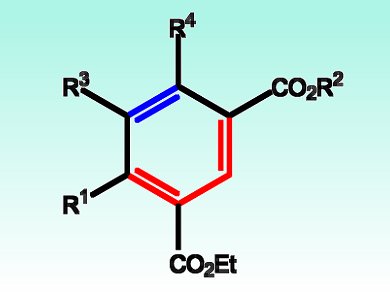
Researchers led by Takuya Kurahashi, Kyoto University, Japan, have developed what can be described as a formal inverse electron-demand Diels-Alder cycloaddition for the synthesis of highly substituted arenes. The team explains that the reaction can be attributed to the formation of an intermediate with the nickel ion, a seven-membered nickelacycle formed from a diene and an alkyne. They point out that activation of both olefins of the diene is essential for the cycloaddition reaction.
Until now, most reactions in this area of transition metal catalysis have used electron-rich dienes.
- Nickel-catalyzed [4+2] cycloaddition for highly substituted arenes,
H. Horie, T. Kurahashi, S. Matsubara,
Chem. Commun. 2012.
DOI: 10.1039/C2CC30801K