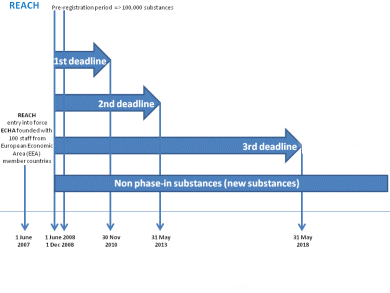
The next Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) registration deadline for substances produced or imported in the EU in quantities at or above 100 t/a is 31 May 2013. The European Chemical Agency (ECHA) organized a Stakeholders’ Day on 23 May to draw the attention of registrants to the timely preparation and quality of their registration dossiers. Video recordings of the event are available for free.
ECHA introduced a new IT-based compliance check strategy which systematically target dossier elements that are immediately relevant for the safe use of the substance. As a consequence, registrants who have submitted non-compliant dossiers may receive multiple decisions from ECHA for a single dossier.
The agency also introduced the substance evaluation process that started in March 2012 and described how it is functionally interlinked with other evaluation and regulatory risk management processes.
An example of a chemical safety report (CSR) was presented. It gives registrants tips for developing a good quality CSR as well as advice on how to avoid common deficiencies.
ECHA also focused on the challenges of applying for authorization and explained which parts of the applications will be made publicly available in order to support interested parties in providing pertinent information on the availability of alternative substances or technologies. And it informed how it will disseminate the opinions of the scientific committees.
Image: enlarge © ChemistryVierws.org
- Video recording of the event
- REACH – An Overview,
ChemViews Magazine 2012.
DOI: 10.1002/chemv.201200029