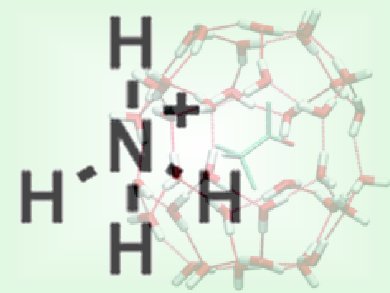
Studies on how ions interact with solvating water molecules could improve our understanding of many processes, particularly of those that take place in the atmosphere and have an influence on climate but also for surface and interface chemistry. Moreover, they will also provide insights into the extent to which ions can influence the hydrogen-bonding network of water.
Evan Williams and colleagues, University of California, Berkeley, USA, have exploited the unique environment within a clathrate cage molecule to investigate the behavior of hydrated, protonated ammonia, various protonated primary, secondary, and tertiary amines and the tetramethylammonium ion surrounded by 19–21 water molecules. Infrared photodissociation (IRPD) spectroscopy and blackbody infrared radiative dissociation (BIRD) at low temperature (133 K) reveal that structural stability does not correlate with the number of hydrogen bonds but that orientation is also important.
The data they obtained should act as useful benchmarks for those working on computational studies of ion effects and hydrogen bonding.
- Locating Protonated Amines in Clathrates,
Terrence M. Chang, Richard J. Cooper, Evan R. Williams,
J. Am. Chem. Soc. 2013.
DOI: 10.1021/ja407414d