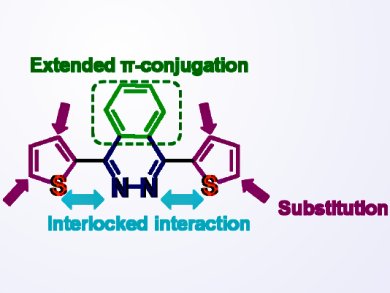
Fan Zhang and colleagues, Shanghai Jiao Tong University, China, synthesized a series of organic conjugated molecules containing 2,3-benzopyridiazine as electron-withdrawing or acceptor (A) core and thiophene derivatives as electron-donating (D) arms.
Ultraviolet-visible (UV-Vis) absorption spectra and fluorescence spectra revealed that the optical properties of these morlecules are strongly influenced by the interactions between nitrogen and sulfur atoms in the conjugated backbone, as well as the position of the alkyl chains in the thiophene rings. The optical properties and electrochemical behaviors of these molecules can be fine-tuned by changing the position of the alkyl chains in the thiophene rings.
The researchers say that their new class of D-A-D conjugated system “will provide some new candidate molecules for application in optoelectronic materials” and that their findings will help to understand the comprehensive effect molecular geometry and electronic structures have on the electronic behavior.
- Synthesis and Physical Properties of Benzopyridazine-Based Conjugated Molecules,
Yuezeng Su, Wei Xu, Feng Qiu, Dongqing Wu, Ping Liub Minzhao Xue, Fan Zhang,
Chin. J. Chem. 2013, 31 (11), 1397—1403.
DOI: 10.1002/cjoc.201300509
The November issue is a Special Issue of Organic Solids


![A Path to Substituted Bicyclo[2.1.1]hexanones](https://www.chemistryviews.org/wp-content/uploads/2024/10/1substitutedbicyclo211hexan2ones_2024-125x94.png)
