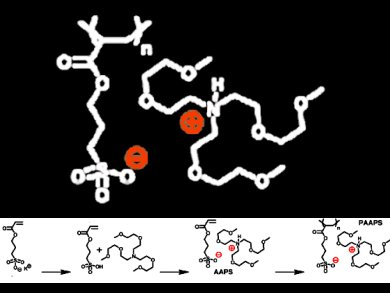
Jiayin Yuan, Max Planck Institute of Colloids and Interfaces, Potsdam, Germany, and colleagues synthesized a poly(ionic liquid) (PIL) via free radical polymerization of 2,2’-azobis(2-methylpropionitrile) (AIBN) and an acrylate-type ionic liquid monomer which was prepared from 3-(acryloyloxy)-1-propanesulfonic acid and tris(2-(2-methoxyethoxy)ethyl)amine.
The PIL, named PAAPS, has a rather low glass transition temperature of −57 °C and exhibits fluidic behavior in a wide temperature range from room temperature to the threshold of the thermal decomposition at 220 °C. Within this range, PAAPS dissolves organic compounds and polymers and separates substances by extraction.
Moreover, the polyelectrolyte serves as reaction medium and stabilizing agent for catalysis and colloid particle synthesis.
The results show that liquid polyelectrolytes have a great potential as solvents in many different reaction systems.
- Polyelectrolyte as Solvent and Reaction Medium,
Simon Prescher, Frank Polzer, Yan Yang, Miriam Siebenbürger, Matthias Ballauff, Jiayin Yuan,
J. Am. Chem. Soc. 2014.
DOI: 10.1021/ja409395y