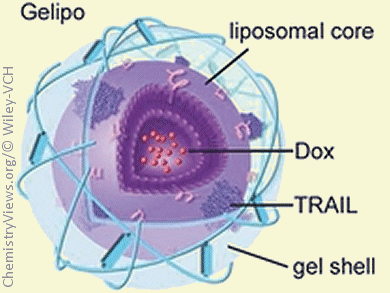
The combination of two or more anticancer drugs induces synergistic effects in tumor cells and, therefore, it is an appealing anticancer strategy. Nevertheless, different drugs have different pharmacokinetic profiles, and thus they may not reach the target tissue with the same kinetics, thereby limiting the efficacy of the combined therapy.
Zhen Gu, University of North Carolina, Chapel Hill, and North Carolina State University, Raleigh, USA, and co-workers reported a way to overcome this problem. The scientists developed a nanoparticle-based delivery platform that simultaneously transports two anticancer molecules. The new system consists of a gel-liposome complex formed by two components. The first one is a liposome-based inner core loaded with the small anticancer molecule doxorubicin. The second one is an outer shell encapsulating TRAIL (tumor necrosis factor related apoptosis-inducing ligand), a therapeutic protein that kills tumor cells. This outer shell is made of crosslinked hyaluronic acid (HA) and it is rapidly degraded once the gel-liposome complex reaches the cancer cells because the tumor environment is rich in HA-degrading enzymes (hyaluronidases). This event, in turn, causes the release of TRAIL and the subsequent internalization of the liposomes into cancer cells. In this way, TRAIL and doxorubicin are simultaneously delivered to tumor cells, thereby killing them through synergistic effects.
- Gel-Liposome-Mediated Co-Delivery of Anticancer Membrane-Associated Proteins and Small-Molecule Drugs for Enhanced Therapeutic Efficacy,
Tianyue Jiang, Ran Mo, Adriano Bellotti, Jianping Zhou, Zhen Gu,
Adv. Funct. Mater. 2013.
DOI: 10.1002/adfm.201303222