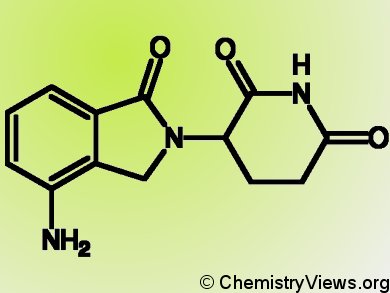
Multiple myeloma is a hematological cancer characterized by the uncontrolled growth of effector B cells, namely antibodies-producing white blood cells. Thalidomide analogs, such as lenalidomide (pictured), are very effective treatments for this disease. Nevertheless, the biochemical mechanism underlying the anti-tumoral activity of these drugs remains unknown.
William G. Kaelin Jr., Dana-Faber Cancer Institute, and Benjamin L. Ebert, Brigham and Women´s Hospital, both Boston, MA, USA, and co-workers demonstrated that lenalidomide induces Ikaros and Aiolos, two important regulators of B cells proliferation and function, to bind to Cerebelon. This molecule is an ubiquitin ligase, namely an enzyme that attaches ubiquitin chains to target proteins, marking them for degradation. Thus, once bound to Cerebelon, Ikaros and Aiolos undergo ubiquitination and subsequent degradation, ultimately causing the death of myeloma cells.
- Lenalidomide Causes Selective Degradation of IKZF1 and IKZF3 in Multiple Myeloma Cells,
J. Kronke, N. D. Udeshi, A. Narla, P. Grauman, S. N. Hurst, M. McConkey, T. Svinkina, D. Heckl, E. Comer, X. Li, C. Ciarlo, E. Hartman, N. Munshi, M. Schenone, S. L. Schreiber, S. A. Carr, B. L. Ebert,
Science 2013, 343, 301–305.
DOI: 10.1126/science.1244851 - The Myeloma Drug Lenalidomide Promotes the Cereblon-Dependent Destruction of Ikaros Proteins,
Gang Lu, Richard E. Middleton, Huahang Sun, MarkVic Naniong, Christopher J. Ott, Constantine S. Mitsiades, Kwok-Kin Wong, James E. Bradner, William G. Kaelin Jr.,
Science 2014, 343 (6168), 305–309.
DOI: 10.1126/science.1244917