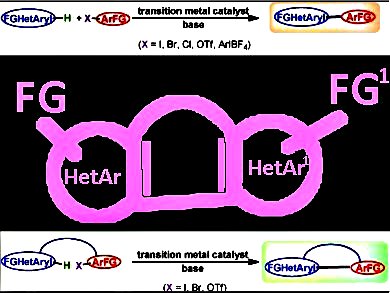
Synthesis and functionalization of heterocyclic compounds is a fundamental segment of academic and industrial research. The development of environmentally friendly and economical methods for the synthesis of heterocyclic compounds is a major priority of this century. One of the most important areas of heterocyclic synthesis is cross-coupling of heteroarenes by C–H functionalization.
Renzo Rossi, Fabio Bellina, and colleagues, University of Pisa, Italy, combine the results of 430 research articles published between January 2009 and February 2013 in the area of the transition metal-catalyzed direct inter- and intramolecular (hetero)arylation reactions of heteroarenes containing one heteroatom with (hetero)aryl halides or pseudohalides. They give particular attention to site and chemoselectivity aspects of these reactions as well as applications of these C–C bond forming reactions in the synthesis of pharmaceutically relevant compounds, natural products, ligands, and photochromic and optical materials.
According to the authors the progress in these reactions has been such that these couplings of (hetero)arenes with (hetero)aryl halides currently can still compete favourably with those in which heteroaryl-(hetero)aryl bonds are formed by transition metal-catalyzed oxidative/dehydrogenative C–H/C–H cross-couplings. In fact, site and chemoselectivity issues, as well as undesired self-coupling pathways, still limit the broad usage of the latter couplings.
They believe that further research efforts are needed to enable the use of transition metal-catalyzed (hetero)arylation reactions of C–H bonds of heteroarenes with (hetero)aryl halides or pseudohalides in industrial processes. Key areas that should be developed include:
- the development of scalable protocols involving very mild reaction conditions and the general use of solvents greener than DMF, DMA, or NMP;
- the design, development, and use of robust, highly efficient, and reliable transition metal catalysts able to work with low or even ultra low loadings. These should be recovered, recycled several times without significant loss of catalytic activity, and may be used for (hetero)arylations of different types of heteroarenes without the use of additives; and
- effective methods for highly chemoselective and site selective (hetero)arylations at the less reactive positions of the heteroarenes, e.g., the C-3 and C-4 positions of furan and thiophene, without the use of blocking groups that are not easily removable at the more reactive positions.
- Cross-Coupling of Heteroarenes by C H Functionalization: Recent Progress towards Direct Arylation and Heteroarylation Reactions Involving Heteroarenes Containing One Heteroatom,
Renzo Rossi, Fabio Bellina, Marco Lessi, Chiara Manzini,
Adv. Synth. Catal. 2014, 356 (1), 17–117.
DOI: 10.1002/adsc.201300922