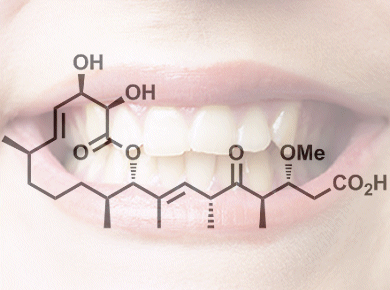
Dental caries, also known as tooth decay, is a global health problem. Although a huge variety of bacteria and pathogens are often present in the mouth, Streptococcus mutans is considered to be a primary agent for tooth decay. Carolacton is a natural product that is active against S. mutans when tested as a biofilm in which cells stick together on a surface, and consequently synthesizing sufficient quantities of carolacton would allow for full biological testing.
William Wuest, Temple University, Philadelphia, PA, USA, and Andrew Phillips, Yale University, New Haven, CT, USA, and colleagues have developed a synthetic route for carolacton (pictured) in which the longest linear sequence is 14 steps and with each step providing moderate to excellent yields (48–99 %). The synthesis begins by using a common β-ketoimide starting material and includes reactions such as macrocycle formation through esterification, ring-closing metathesis, and selective hydrogenation to generate the product. The synthesized carolacton was then tested against S. mutans as a biofilm, and dramatic effects were seen.
Work will now focus on further understanding how carolacton and similar compounds interact with bacteria, and the exact mechanism of action with S. mutans in biofilms.
- A Concise Synthesis of Carolacton,
Michal S. Hallside, Richard S. Brzozowski, William M. Wuest, Andrew J. Phillips,
Org. Lett. 2014.
DOI: 10.1021/ol500004k