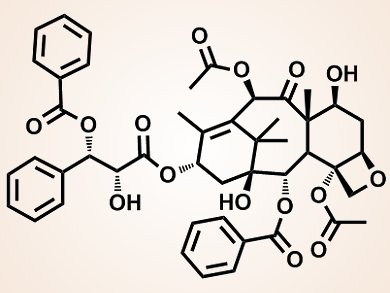
The treatment of glioma, a highly aggressive brain tumor, is very challenging. Chemotherapeutic drugs often fail to reach the cancerous cells because of the blood–brain barrier, a semipermeable barrier separating the blood circulation from the nervous tissue. The drugs instead reach other organs and this can cause side effects to occur.
Chen Jiang and co-workers, Fudan University, Shanghai, China, developed smart nanodevices that efficiently permeate glioma cells. Firstly, using the amphiphilic polypeptide copolymer PEG-pLys-pPhe, the scientists fabricated polymeric micelles that were then loaded with dehydroascorbic acid (DHA). In this way, the researchers obtained glioma-targeting micelles as DHA rapidly diffuses into glioma cells and is subsequently trapped in them. Next, the micelles were cross-linked to the chemotherapeutic drug paclitaxel (pictured) by disulphide bonds.
As the intracellular levels of glutathione are very high in tumor cells, once the micelles are internalized in glioma cells, the disulphide bonds linking paclitaxel to the micelles are cleaved and paclitaxel released. Thus, in addition to selectively targeting glioma cells, these micelles release the chemotherapeutic drug in a controlled manner, preventing its leakage into healthy tissues. Consequently, they reduced the growth of the glioma without causing toxic side effects.
- Smart Nanodevice Combined Tumor-Specific Vector with Cellular Microenvironment-Triggered Property for Highly Effective Antiglioma Therapy,
Kun Shao, Ning Ding, Shixian Huang, Sumei Ren, Yu Zhang, Yuyang Kuang, Yubo Guo, Haojun Ma, Sai An, Yingxia Li, Chen Jiang,
ACS Nano 2014, 8, 1191–1203.
DOI: 10.1021/nn406285x