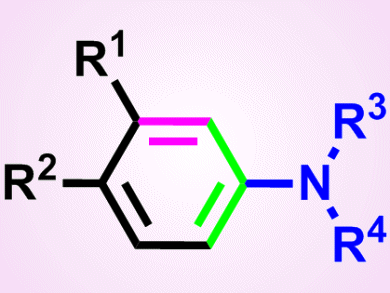
Aryl amines are present in many organic chemistry areas including medicines, materials and natural products, and there are a variety of ways in which they may be prepared. However, for a number of these routes, the aryl fragments need to be functionalized prior to reaction, which restricts their range. Consequently, new strategies that avoid this problem would allow for a greater range of functional groups to be used, and may also allow access to aryl fragments with unusual substitution patterns.
Timothy Donohoe and colleagues, University of Oxford and AstraZeneca Pharmaceuticals, Macclesfield, both UK, have developed a new strategy based on an olefin cross-metathesis reaction, a reaction in which two starting materials containing C=C bonds (olefins) react together to give an intermediate (a 1,5-dicarbonyl). Onward reaction of the intermediate, which goes on to form the aryl fragment, with either a primary or secondary amine, leads to the aryl amine. A wide range of intermediates, containing arene, alkane, and halogens, reacted well with amines, such as piperidine and simple alkyl amines, to give the products in good yields, typically 60–90 %.
- De Novo Synthesis of Multisubstituted Aryl Amines Using Alkene Cross Metathesis,
Matthew R. Tatton, Iain Simpson, Timothy J. Donohoe,
Org. Lett. 2014.
DOI: 10.1021/ol500441q


![A Path to Substituted Bicyclo[2.1.1]hexanones](https://www.chemistryviews.org/wp-content/uploads/2024/10/1substitutedbicyclo211hexan2ones_2024-125x94.png)
