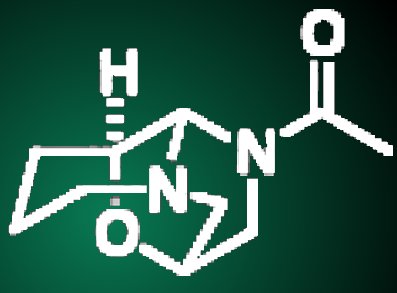
Lepistine is an organic alkaloid compound that is a major component found in the mushroom Clitocybe fasciculate. This compound is made up of three attached rings, contains two nitrogen atoms and one oxygen atom in the carbon skeleton, and forms a closely folded structure. Although lepistine was first isolated in 1975, and despite its unusual structure, no synthetic or biological studies on the compound were performed.
Intrigued by the compound, Satoshi Yokoshima, Tohru Fukuyama, and colleague from Nagoya University, Japan, have developed a synthetic route that features an easy way to form the tricyclic skeleton. By starting from a small compound called (S)-glycidol, which contains an alcohol group and an epoxide (a three-membered ring containing an oxygen atom), the synthesis of lepistine was achieved over 11 reaction steps. The reaction pathway involves the formation of a 10-membered single-ring compound. This large single ring is then bridged (enol ether), which results in two smaller rings. A subsequent intramolecular reaction (aminal formation) forms the third and final ring.
- Total Synthesis of (−)-Lepistine,
Yusuke Kitabayashi, Satoshi Yokoshima, Tohru Fukuyama,
Org. Lett. 2014.
DOI: 10.1021/ol5010033