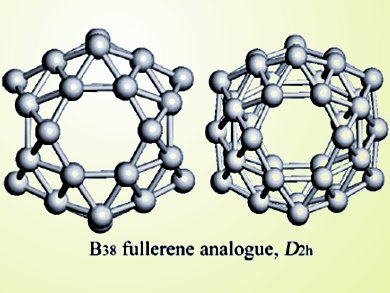
The exotic structure and properties of C60 fullerene has inspired chemists to seek its analogues made of elements other than carbon. Boron features the B12 icosahedron motif in bulk materials, and can form many types of multi-atom clusters, giving it great potential to form fullerene-like structures.
Through first-principles calculations, Yanming Ma and co-workers at Jilin University, Changchun, China, have identified a stable D2h-symmetric boron cage composed of 38 atoms. Using an algorithm optimized for cluster prediction with a global minimization of energy surface, the authors found this highly symmetric B38 fullerene analogue made of 56 triangles and 4 hexagons, stabilized by a large energy gap and σ and π dual aromaticity.
The newly revealed medium-sized boron cluster is predicted to be more stable than the common quasi-planar B12 and double-ring B20 clusters. In addition, these findings may eventually lead to the experimental synthesis of this B38 fullerene analogue.
- B38: An All-boron Fullerene Analogue,
J. Lv, Y. Wang, L. Zhu, Y. Ma,
Nanoscale 2014.
DOI: 10.1039/C4NR01846J