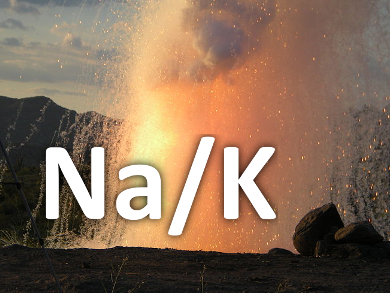It is high school chemistry knowledge that alkali metals, such as sodium and potassium, react violently upon contact with water, yielding sodium hydroxide and hydrogen, and may even blow up. The explosion is usually believed to be caused by the massive heat released during this very fast process.
However, evidence captured by Pavel Jungwirth and co-workers, Academy of Sciences of the Czech Republic, Prague, and Technische Universität Braunschweig, Germany, who used high-speed cameras to look at the early stage of the reaction, may disprove this long-held interpretation. Following the process of dropping a liquid Na/K alloy in water, the researchers observed metal spikes growing out of the metal surface within a submillisecond timeframe, which repeats upon formation of new metal surfaces. As suggested by molecular dynamics simulations, the immediate electron transfer from Na and K to water overcharges the alloy droplet, leading to its rapid disintegration as a result of strong coulomb repulsion, a phenomenon called ‘coulomb explosion’.
- Coulomb explosion during the early stages of the reaction of alkali metals with water,
Philip E. Mason, Frank Uhlig, Václav Vaněk, Tillmann Buttersack, Sigurd Bauerecker, Pavel Jungwirth,
Nat. Chem. 2015.
DOI: 10.1038/nchem.2161